
Expired activity
Please go to the PowerPak
homepage and select a course.
Opioid Analgesics: Best Practices for Prescribing, Dispensing, and Preventing Diversion
INTRODUCTION
To understand West Virginia's prescription drug abuse problem–often described as epidemic in proportion and exploding in magnitude–pharmacists need to understand specific trends and realities both at the national level and in West Virginia (WV). Epidemiologists noted sharp increases in nonmedical use of opioid analgesics between 2002 and 2011. Recreational drug users and addicts often use benzodiazepines as companion drugs when they misuse opioids, so they are part of the problem.1 Although the problem seems to have stabilized or declined in some areas, it is still a public health crisis.
This continuing education program walks pharmacists through prescription opioid abuse in an organized way. It starts by discussing the statistics and terminology. It then covers the motivating factors–the reasons why prescription opioid abuse began and became attractive to recreational and addicted drug users. Next, it discusses the players–the people who have roles in this crisis. It covers how addicts and abusers obtain and use these drugs. Finally, it covers solutions to reduce harm and treatments to help those who are already addicted.
EPIDEMIOLOGY OF CHRONIC PAIN AND OPIOID ABUSE
In 2012, opioid prescribing reached a peak in the United States, with American physicians writing more than 255 million prescriptions for these agents, a prescribing rate of 81.3 per 100 persons. This has since declined to 191 million (58.7 prescriptions per 100 persons), according to 2017 data. West Virginia ranked third in the country in 2012 in prescribed opioid usage, with 137.6 prescriptions per 100 persons. By 2017, this rate had declined to 81.3 opioid prescriptions per 100 persons; while still above the national average and in the top 10 states in terms of usage, the decline showed substantial progress in addressing the opioid crisis in West Virginia. However, this rate fails to capture the use of illicit opioids, a problem that has worsened during this time period.2
West Virginians are more likely to die of drug overdoses than auto accidents, but this is not unique to WV–people in 28 other states also have higher rates of deaths from drug overdoses than auto accidents.3 Data indicate WV's miners, poor, and rural populations are affected by opioid abuse disproportionately.4 It's time to look at the problem closely and critically, and take action.
Public health officials look at prescription drug abuse from many angles to gain insight. The Centers for Disease Control and Prevention (CDC) surveys the population concerning past 30-day (recent) use of opioid analgesics as one indicator of change. Among Americans aged 20 and older, recent use increased during 1988–1994 (3.4%), 1999–2002 (5%) and 2003–2006 (6.9%), but stabilized during 2007–2010 (6.7%) and 2011–2012 (6.9%).5 Table 1 describes past use during a longer period–1 year–and indicates that West Virginians seem to be using opioid analgesics less, but the problem is still substantial.
| Table 1. Past-year nonmedical opioid analgesic use, comparing 2002–03 and 2012-13 |
| Age group |
Nationwide |
West Virginia |
| 2002–03 |
2012–13 |
2002–03 |
2012–13 |
| Ages 12+ |
4.79% |
4.51% |
5.34% |
3.84% |
| Ages 12–17 |
7.61% |
5.00% |
8.87% |
4.16% |
| Ages 18–25 |
11.70% |
9.47% |
15.67% |
8.65% |
| Ages 26+ |
3.20% |
3.60% |
3.27% |
3.07% |
| Source: Reference 6 |
Other indications of opioid threat
Other national statistics serve to emphasize this problem's scale:
- From 1999 to 2013, annual rates of poisoning deaths more than doubled for all drugs (6.1 to 13.8/100,000 population), but almost quadrupled for opioid analgesics (1.4 to 5.1/100,000). Both showed greater increases in 1999–2006 than 2006–2013.7
- In 2013, opioid analgesics were involved in 37% fatal drug poisonings.
- Benzodiazepines contributed to 13% of opioid deaths in 1999 and 31% in 2011.1 Alcohol was a contributing factor in 20% of opioid deaths in 2010.8
In WV, opioids were the leading cause of drug-related deaths, increasing 550% from 1999 to 2004 (largest state increase in the United States) and 214% from 2001 to 2010. WV also had the highest age-adjusted rate of opioid-related deaths in 2012 and the highest death rate involving prescription drugs in 2008.9,10 From 2009 to 2012, opioid exposure incidents reported to the WV Poison Control Center increased by 12.5%.9 Statistics are basically numbers. For health care professionals, they provide some evidence that the opioid problem is real, even if their source or accuracy is questioned. For patients, statistics have little meaning. They are more concerned about the reality of the problem–the addict in the family; the social, emotional and economic cost of continuing to abuse drugs; and the reality that abuse is risky.
Definitions of terms
The opioid abuse problem–classified as a psychiatric illness–can be confusing, even to experts. Much of the confusion stems from the large number of definitions, many of which are used inconsistently. Table 2 lists common terms.
| Table 2. Behavioral responses to prescribed opioid analgesics |
| Term |
Definition |
| Misuse and nonmedical use |
Medication use that is different than what the prescriber intended |
| Abuse |
A maladaptive pattern of opioid use with the primary intent of achieving euphoria or getting high |
| Addiction |
Addiction (characterized behaviorally and not physiologically) is a persistent, compulsive pursuit of a behavior or substance to find relief or reward |
| Physical dependence |
Drug use that causes specific withdrawal syndrome following abrupt cessation or dose reduction, decreased blood concentration, or antagonist administration |
| Tolerance |
A state of adaptation in which the physiologic changes from drug exposure over time lead to diminished drug effect |
| Iatrogenic |
Medical or surgical treatment-induced morbidity |
| Pseudo-addiction |
An iatrogenic condition where patients display aberrant drug-seeking behaviors mimicking opioid use disorder, but driven by intense need for pain relief. Resolves with adequate pain control |
| Diversion |
Transfer of a controlled substance from authorized to unauthorized possession or distribution |
| Source: References 11-13 |
Most clinicians use the American Psychiatric Association's (APA's) Diagnostic and Statistical Manual (DSM) to diagnose psychiatric conditions. Acknowledging that criteria used previously were prone to misinterpretation, the 2013 DSM-5 merged the criteria for "substance dependence" and "substance abuse."14 The DSM-5 eliminated physiologic dependence as a diagnostic criterion. The DSM-5 refers to substance abuse and dependence as substance use disorder (SUD) and combined opioid abuse and dependence as opioid use disorder (OUD), with severity of both measured on a continuum.14
The American Society of Addiction Medicine defines addiction as…"a primary, chronic disease of brain reward, motivation, memory and related circuitry. Dysfunction in these circuits leads to characteristic biological, psychological, social and spiritual manifestations. This is reflected in an individual pathologically pursuing reward and/or relief by substance use and other behaviors. Addiction is characterized by inability to consistently abstain, impairment in behavioral control, craving, diminished recognition of significant problems with one's behaviors and interpersonal relationships, and a dysfunctional emotional response to the substance..." People's addictions manifest with unique symptoms and in a range of severities.15
Factors driving opioid abuse
Three issues contributed to the current opioid abuse problem: medical, social and economic. When combined, these issues created a problem that is vastly different than past illicit drug epidemics. The situation has resulted in considerably more deaths than past heroin and crack cocaine epidemics, and has been much more difficult to address. The first driver was change in the medical establishment's view of pain and its treatment. Until the 1980s, physicians believed that only cancer and postoperative pain warranted the strongest of opioids. Medical opinion then changed. Experts argued that all forms of chronic pain needed more aggressive treatment.16 By 2000, Congress adopted a similar position, declaring the new century's first decade the "decade of pain control and research."17 Accrediting agencies promoted pain as the fifth vital sign, and campaigns to ensure that patients with chronic noncancer pain ensued. Addiction risk was downplayed (when OxyContin was introduced to the market in 1996, its product information stated the addiction risk was <1%18). Advertising proliferated. By 2002, doctors were prescribing 10 times more OxyContin than they had in 1997.
At the same time, patients were invited to become partners in their care, and patient satisfaction scores were introduced into medical practice. Pain is not objectively measurable (see Sidebar). It is a subjective sensation that only the patient can describe. Many organizations linked physicians' pay to patient satisfaction, creating pressure on prescriber to provide analgesics when patients said they needed them.19-21 The line between suffering and drug-seeking behavior became blurry.
In addition, for some patients, prescription opioids represented an improvement over street drugs. They were cleaner drugs that delivered a predictable high with a better margin of safety. Abusers and addicts fall victim to an illusion of safety and legitimacy; prescription opioids were at least consistent in the amount of drug delivered. However, illicit drug use seems to have more associated stigma, and individuals may be more likely to initiate experimentation with "safe" prescription drugs.22
The economics of the situation has been and remains quite complex. Factors that influenced opioid use include direct marketing to consumers; involvement of physicians and pharmacists; enhanced, bonus-driven drug sales forces; and Mexican drug cartels' recognition that appropriate and inappropriate opioid use was exploding. "Pill mills"– physician-staffed facilities that require little medical documentation, treat pain liberally with medication, often allow patients to select their own opioid, and operate on a cash- only basis–began to appear in many areas. As law and public health officials scrambled to close them, cartels and drug dealers were poised to service patients addicted to but unable to access opioids. Heroin made a comeback, and it is cheaper and more addictive than prescription opioids.23 Opioid abuse–even among people who start using opioids for real pain–became an entryway to abuse of harder, illicit drugs.24
Nationwide, total treatment admissions for nonheroin OUD increased 348% from 2001 to 2011, with prevalence increasing each year.25 The proportion of drug treatment admissions for nonheroin OUD in WV increased from 16% in 2003 to 59% in 2010.26
|
Pain: The basics
Pain, a complex phenomenon, has been explained by several models.
The traditional biomedical model assumes a causal relationship between a specific pathophysiologic process and pain presence and severity. Pain is a symptom that resolves by treating the underlying injury or disease. While valid for acute pain, this model ignores psychosocial contributions to pathogenesis, making it less useful in chronic pain assessment and treatment.19-21
The biopsychosocial model recognizes that pathophysiology, psychologic status, cultural background/belief system, and relationships and interactions in workplace, home, and health care environments influence pain. In chronic pain, the sensation perceived as pain becomes uncoupled from its original cause. Pain persists long after tissue healing because structural and functional alterations in the peripheral neurons and central nervous system (CNS) amplify and perpetuate the sensation.19-21
To manage chronic pain effectively, prescribers and pharmacists must replace disproven assumptions with recent knowledge advances. These define pain as follows27:
- An inaccurate measure of tissue state
- Modulated by multiple physiological, psychological, and social factors
- Less reliant on the pain–tissue state relationship if it becomes chronic
- Present even if imaging or diagnostic intervention do not confirm a cause
This last point is important. Some pain practitioners and organizations perpetuate the myth that unless pain complaints are objectively confirmed, the patient is probably drug seeking. Chronic pain often cannot be objectively confirmed.
|
Opioid manufacturers, prescribers, and dispensers
Prescription opioids are federally scheduled narcotics. As such, these drugs originate in pharmaceutical manufacturing facilities and make their way to pharmacies through distributors. All manufacturers, distributors, physicians, pharmacies, and others in the controlled substance supply chain are required to register with the DEA (unless exempt) and maintain strict accounting of all controlled substance transactions.28 Those in the opioid product supply chain have a cradle-to-grave responsibility for drugs, meaning that the parties should be able to track drugs from their manufacture to their environmentally safe disposal.
How do opioids seep into illicit and recreational circulation from the closed U.S. Drug distribution system? Pharmaceuticals making their way outside this closed system are termed "diverted."12,29 Most opioid diversion occurs after prescriptions are dispensed.30 Health care professionals prescribe, dispense, and administer prescription opioids, serving as middlemen, gatekeepers, and monitors of appropriate use. The various factors discussed above have caused our system to fail. It is time to step up our efforts and patch holes in our distribution and prescribing systems, and look at all stakeholders' demographics and risk factors.
Health care professionals
Prescribers influence prescription opioid abuse most heavily.22 Five behaviors in particular appear to be linked to increased opioid availability:
- Prescribing more opioids, especially for chronic noncancer pain; increasing opioid use for chronic noncancer pain correlates with increasing overdose and more emergency department visits.
- Prescribing opioid doses that often exceed those recommended by clinical practice guidelines.
- Prescribing oxycodone, especially in high doses and in the long-acting form.
- Employing methadone for pain relief (not as a substance abuse treatment) often to save money but without considerations of its dangerously narrow therapeutic window.18
- Prescribers tend to over-rely on equianalgesic dose tables for opioid conversion, especially involving methadone.31
- Writing a large volume of prescriptions, which decreases the quality of patient assessment.
Pharmacists have a pivotal, if somewhat uncomfortable role, in this epidemic. The regulatory response has been sweeping, complicated, and inflexible. Prescribers often resent or berate pharmacists who try to intervene when they suspect inappropriate opioid prescribing or patient abuse or addiction. Prescribers often allege that pharmacists' queries challenge their professional judgment and take them away from patient care.32 However, pharmacists who monitor prescribing closely and question situations that seem implausible improve patient care and ensure safe drug use. Experts recommend that pharmacists use noninvasive means, such as database inquiries, before contacting prescribers. As this epidemic continues and more health care professionals embrace control measures, collaboration will be critical.32
Patients
Opioid analgesics may relieve physical and psychological pain, emotional distress, negative emotional states, and insomnia. Misconceptions are widespread about opioids and abuse. For example, some people refer to opioids as "addictive drugs." Opioids are not necessarily addictive, but they do have abuse potential.33,34
Most people who take drugs with high abuse potential use them as prescribed and without addiction.33,34 People who are highly susceptible to opioid reward effects experience euphoria and a sense of well-being.35 For example, patients with chronic pain who develop OUD appear to respond to opioids with euphoria, stimulation, and other subjective experiences; this is distinctly different from patients who experience pain relief without developing OUD.36
Similarly, patients respond to and tolerate opioid analgesics differently. Patients who respond incompletely may experience inadequate pain relief, emerging OUD, or both. Pseudo-addiction describes problematic behaviors specifically driven by desperation over unrelieved pain.20 These patients are driven by real pain that persists at their prescribed dose. They may complain, ask for higher or more frequent doses, openly obtain opioid analgesics from alternative sources, deceive prescribers, stockpile unused medication, and self-adjust their doses upward. Pseudoaddictive behaviors resolve once these patients have adequate pain control.37,38
Abusers or addicts
Pain patients' problematic behaviors are often diagnosable as OUD, but nonmedical use/misuse of opioid analgesics is also a driver. Psychiatric disorders that cause impulsive drug use, unresolved family issues, cognitive disorders, or criminal intent can also create addicts. Motive and risk varies, and every abuser and addict has a unique set of circumstances.
Patient risk factors for prescription opioid abuse include age under 25 years; patient or family history of alcohol or substance use disorder; past or current psychiatric disorder; cigarette smoking; and patient history of physical, emotional, or sexual abuse, aberrant drug-related behaviors (ADRB, listed in Table 3, column 1), or legal problems. Importantly, evidence indicates that high-risk patients (those with multiple risk factors) do not necessarily develop ADRB or OUD during opioid analgesic treatment, and that ADRBs develop at much higher rates than OUD.19,39,40 Alcohol use disorder (AUD) and OUD frequently co-occur, and nonmedical opioid use increases in tandem with alcohol consumption. People with pre-existing AUD have 20 times the risk of developing OUD compared with people without AUD.30
Some people's behaviors and characteristics increase their risk of death from opioid overdose. Patients increase their chances of overdose when they take diverted opioids (prescribed for someone else), doctor- or pharmacy-shop, are polydrug users (especially involving benzodiazepines), or indiscriminately substitute one drug for another. Of note, a study conducted in WV found that patients who doctor-shopped were 6 times more likely to die, and pharmacy-shoppers were 13 times more likely to die of an overdose than an abuser who did not.41
Distinguishing legitimate patients from drug abusers is not easy, but drug-seeking abusers and doctor-shoppers often share similar traits and behaviors (Table 3). Recognizing these characteristics may help pharmacists identify manipulative individuals.
| Table 3. Behaviors associated with drug seeking and diversion |
| Behaviors suggesting drug-seeking |
Behaviors suggesting possible diversion |
- Unusual behavior
- Unusual appearance (unkempt or overdressed)
- Overly assertive
- Knowledge of controlled substances beyond that of the typical lay person
- Claims to have no regular doctor and no health insurance
- Is not a resident of the city or town; alleges to be in town to travel or visiting friends/relatives
- Claims prescription was lost or stolen; requests replacement
- Requests refills more frequently than prescribed
- Manipulates through sympathy, guilt, or direct threats; uses a child or older person to obtain methylphenidate or opioids
- Cutaneous signs of drug abuse with skin tracks or scars on neck, arms, feet, or ankles
|
- Known contact with drug-abusing persons or groups
- Nonadherence to other treatment recommendations
- Preference for drugs with high street value
- Insistence on brand-name drugs
- Negative urine screens when the prescribed opioid or metabolites should be detectable
|
| Adapted from references 42 and 43. |
ADRBs sometimes suggest emerging OUD. Closer assessment is required, as several ADRBs are similar to signs of pseudoaddiction. ADRBs that are more predictive of OUD include opioid tampering for intranasal or IV use, prescription forgery, selling opioids, stealing opioids, street purchase of prescription or illicit drugs, and repeatedly losing opioid prescriptions.44
Sources and methods of abuse of opioids
It's reasonable to wonder how, in a system that runs on accountability, abusers obtain opioid analgesics, and how they use them.
Opioid sources
Findings from a CDC study indicate that most people (54.4%) who abuse prescription opioids get them for free from a friend or relative. Another 16.2% also get opioids from friends or relative, but buy or steal them. All other sources pale in comparison. However, the people who are most likely to overdose (27% of patients surveyed) are those possessing their own physician-provided prescriptions. High-risk users also do not restrict themselves to opioids obtained on their own prescriptions or drugs from friends and family–they are 4 four times more likely than the average user to buy the drugs from dealers or strangers.45
For this reason, ideal prevention efforts focus on physicians' prescribing behaviors, pharmacy dispensing data, and patients at highest risk for overdose. Ideally, health care providers would screen for abuse risk and check state prescription drug monitoring programs before prescribing. Collecting unused medications through take-back events is also a way to shut down this avenue of diversion.45
Abuse methods
Many abusers and addicts use unaltered prescription opioids, taking them orally or using transdermal patches. Hard-core abusers typically graduate to inhaling, smoking, or intravenous use. By tampering with dosage forms (chewing, crushing, or dissolving the dosage form), they create a more concentrated drug that gives them a faster or more profound high.46
Many addicts are well informed about drugs and formulations. They alter the dosage form to remove undesirable ingredients or make the drug easier to administer by a different route. Directions for drug tampering are easily found on the Internet.46 Following OxyContin ER's 1996 introduction, users learned to crush and swallow, sniff or snort, or dissolve the powdered tablet for injection to produce a very intense euphoric drug effect. ER-opioid tampering increased through the late 1990s and 2000s, greatly elevating risks of overdose and development or exacerbation of OUDs.30,47
Specific drugs
Opioids' potential to cause euphoria, common to all opioid analgesics, attracts abusers. While addiction is largely related to patient factors, abuse potential is largely shaped by reward intensity. Reward intensity is influenced by the specific opioids' pharmacodynamic activity and the formulation's pharmacokinetic characteristics.42 Greatest reward intensity is associated with pure mu opioid receptor (MOR) agonists (morphine, oxycodone, hydrocodone). Partial MOR agonists (buprenorphine) and mixed MOR agonist/antagonists (pentazocine, butorphanol, nalbuphine) are more likely to produce dysphoria, especially with higher doses. Their lower abuse potential is due in part to increasing side effects (dysphoria, hallucinations) with dose escalation, not by analgesia or reward.19,42
Opioid formulations' pharmacokinetic properties, such as maximum plasma concentration (Cmax) and time to reach Cmax (Tmax), also influence abuse potential. Drugs with comparatively short Tmax have higher abuse potential. Their rapid elevation in the plasma and CNS produce greater euphoria and "rush" than drugs with slower onsets. Patient's tolerance level influences reward because identically dosed opioids may produce euphoria in the opioid-naïve individual but have no effect in the opioid-tolerant person.44,42 Opioid users also have preferences including duration of effect, route of administration, and formulation.48
In the real-world setting, abuse potential, potency, and duration of action do not necessarily translate into or predict abuse prevalence; price, availability, and previous exposure may have greater relevance.48,49 Abuse potential can be inferred from street price, pharmacy theft rates, drugs leading to treatment access, and overdose morbidity and mortality data. Substantially more hydrocodone than oxycodone tablets were sold in 2011, but oxycodone was disproportionately represented in emergency department visits for nonmedical use. This suggests oxycodone is preferred for recreational use. Raw numbers of abuse cases or events require adjustment to compare abuse potential accurately across opioids.49
As noted above, LA oxycodone and methadone appear to be the most dangerous of the opioids.22 Oxycodone's risk is based primarily on its place as the preferred opioid among abusers. Approximately 45% of abusers prefer oxycodone, citing the quality of the high. Risk-tolerant young men who prefer to inject or snort their drugs to get high and are willing to use more aggressive forms of diversion are most likely to prefer oxycontin, according to one study.24
Approximately 30% of abusers prefer hydrocodone, although many indicate that inclusion of acetaminophen limits dose escalation necessary for "getting high." Hydrocodone users tend to be risk-averse women, older people, noninjectors, and those who prefer safer modes of acquisition than dealers (i.e., doctors, friends, or family members).24
Relatively low cost has been a driving factor in use of methadone for pain. Inexperienced prescribers who underestimate methadone's complex pharmacology play with fire. By 2009, methadone accounted for 2% of opioid analgesic prescriptions and more than 30% of opioid analgesic deaths.50
Mortality with other analgesics
Importantly, nonopioid analgesics cause fatalities. Acetaminophen and nonsteroidal anti- inflammatory drugs are responsible for 15.3 deaths per 100,000 users annually from fatal gastric, hepatic, and renal toxicities. Unlike these analgesics, opioids are not associated with fatal end-organ toxicity; death results from acute respiratory suppression culminating in cardiac arrest.19 Many short-acting (SA) opioids are combined with acetaminophen or NSAIDs and now have a dose ceiling to avoid nonopioid toxicity.44
Opioid laws and regulations
Although the 1970 Controlled Substances Act (CSA) was introduced to reduce abuse and diversion, its creation of the Drug Enforcement Administration (DEA) and that agency's regulations have been insufficient to control this epidemic.
Pharmacists have a professional responsibility to protect their practices from becoming easy targets for drug diversion. This starts with a constant vigilance against forged or altered prescriptions. The CSA holds pharmacists personally responsible for knowingly dispensing controlled substances (CS) outside of the usual course of professional treatment. Especially with regard to opioid prescribing for pain, prescribed drug quantity and prescription filling frequency should not be used as sole indications of improper prescribing or prescription fraud.
Tables 4–6 summarize basic information about CS prescribing.
|
Table 4. Legal requirements for prescriptions for controlled substances (CS)28
- Dated and signed on the date issued by the prescriber
- Patient's full name and address
- Practitioner's full name, address, and DEA registration number
- Drug name
- Strength
- Dosage form
- Quantity prescribed
- Directions for use
- Number of refills (if any) authorized
- Written in ink or indelible pencil, or typewritten
- Prescribers must manually sign and date the prescription, but can delegate prescription preparation to other personnel.
- Schedule II prescriptions must be written only, with no refills allowed. Oral order permissible only in emergency situation, and facsimile (fax) prescriptions allowed only in certain situations (compounding for administration to patient; long-term care facilities; hospice care programs).
- Schedule III, IV, and V prescriptions can be written, oral, or sent by facsimile; up to 5 refills within 6 months allowed.
|
|
Table 5. Verifying DEA numbers28
- DEA registration numbers for physicians, dentists, veterinarians and other practitioners start with the letters A, B, or F. Those for midlevel practitioners begin with the letter M.
- The first letter is almost always followed by the first letter of registrant last name, and a computer-generated sequence of 7 numbers. For example, a midlevel practitioner with last name Jones may have the DEA number MJ3614511.
- Practitioners employed by hospitals or other institutions may administer, dispense, or prescribe controlled substances under the registration of their employer and forego individual registration. For the sample DEA number AB1234567–012, the first 9 characters are the hospital DEA registration number, and the last 3 numbers are the prescriber's hospital code number.
|
| Table 6. Types of fraudulent prescriptions and indicators of illegitimate purpose28 |
| Types of fraudulent prescriptions |
Indications that a prescription was not issued for a legitimate medical purpose |
- Altered legitimate prescription
- Prescription pads printed using a legitimate doctor’s name, but different number answered by an accomplice for call-back confirmation
- Some may call in their own prescriptions, using their own phone number for call-back confirmation
- Stolen blank prescription pads from practitioner’s offices, prescription written using fictitious patient names and addresses
- Emergency department visit for pain, opioid prescription received is altered or copied to use again
Signs of a forged prescription
Appears:
- “Too good,” handwriting too legible
- “Textbook”
- Photocopied
- Written in different color inks or handwriting
- Departs from standard medical use in:
- Quantities
- Directions
- Dosages
- Abbreviations
- No abbreviations, written in full
|
- Prescriber writes significantly more prescriptions (or in larger quantities) than is normative for the region.
- Patient appears to return too frequently: A prescription that should last a month in legitimate use is refilled biweekly, weekly, or daily.
- Prescriber simultaneously writes prescriptions for drugs with opposite actions, such as CNS depressants and stimulants.
- Patient presents multiple prescriptions written for other people.
- Several people appear together or within a short period, producing similar prescriptions from the same physician.
- Several people who are not regular customers or community residents appear with prescriptions from the same physician.
|
| Source: Reference 28 |
Tools for addressing opioid misuse and abuse
The extent to which controlled substances are abused or sold illegally is greatly influenced by how readily specific drugs are diverted from legitimate distribution.12,29 Lawmakers, public health officials, health professionals, and concerned citizens have developed a number of tools to augment CSA. What do pharmacists need to know about these tools?
Best practices for prescribers and pharmacy professionals
Some tools are available for health professionals to use routinely in practice. Some of these represent standard and routine practice, while others are specific to opioids.
Careful assessment and awareness
The best way health professionals can prevent opioid abuse is careful assessment and awareness. Before initiating opioids, prescribers should know how to initiate therapy, modify/titrate/taper doses, discontinue use, and apply opioid tolerance criteria (use of some ER/LA products/doses is restricted to opioid-tolerant patients). Prescribers must also know the symptoms of opioid intoxication, overdose, and withdrawal.
Prescribers. In initiating therapy, prescribers should assess the patient for opioid misuse/abuse risk factors. These include a patient or family history of SUD or a patient history of psychiatric illness. Pregnancy status, current medications, and abuse histories of household contact should also be considered.39,51
Before initiating therapy, prescribers should be familiar with ER/LA opioid analgesics, including the following specifics51:
- ER/LA opioid analgesics should be reserved for patients in whom nonopioid analgesics or SA opioids are ineffective, not tolerated, or likely inadequate for sufficient pain management.
- ER/LA opioid analgesics are contraindicated in patients with significant respiratory depression or acute/severe asthma unless administered in a monitored setting or a location where resuscitative equipment is present; paralytic ileus; and/or hypersensitivity.
- Fatal respiratory depression is a potential risk of all ER/LA opioids, and it is further increased by crushing, chewing, or dissolving an oral dose unit; accidental nonpatient ingestion, especially by children; and alcohol or benzodiazepine co- ingestion. QT interval prolongation and torsades de pointes may occur with buprenorphine or methadone.
- Neonatal opioid withdrawal syndrome (NOWS) can occur with prolonged ER/LA opioid analgesic use during pregnancy, and the condition is possibly life- threatening if not recognized and treated. If the drugs are continued during pregnancy, the prescriber should advise the mother about the NOWS risk and ensure treatment availability at birth of the child.
- Constipation is the most common side effect of ER/LA opioid analgesics; others include nausea, vomiting, sedation, and dry mouth.
- Initiating CYP3A4 inhibitors or discontinuing CYP3A4 inducers can result in fatal overdose during ER/LA buprenorphine, fentanyl, methadone, or oxycodone therapy by increasing plasma opioid concentrations.
- Adverse drug interactions can occur during concomitant therapy with ER/LA opioids plus monoamine oxidase inhibitors, anticholinergic agents, skeletal muscle relaxants, and/or mixed agonist/antagonist or partial agonist opioids.
During ongoing treatment, prescribers should know how to manage ongoing ER/LA opioid therapy. This includes:
- Routine monitoring of safety, efficacy, abuse, or addiction
- Regular, documented evaluation of pain severity and impact, progress in functional goals, adverse events, and ADRBs
- Use of prescription drug monitoring programs (PDMPs) and urine drug testing for adherence monitoring
The American Academy of Pain Medicine (AAPM) provides good advice to health professionals regarding use of opioids for treatment of chronic pain. The organization suggested the following when chronic opioid therapy (COT) is used52:
- Legislation and regulatory policies should limit inappropriate prescribing but should not discourage or prevent prescription of opioids where medically indicated and appropriately managed.
- Prescription of opioids for chronic, intractable pain is appropriate when more conservative methods are ineffective and the treatment plan is reasonably designed to avoid diversion, addiction, and other adverse effects.
- Physicians should be sensitive to and seek to minimize the risks of addiction, respiratory depression and other adverse effects, tolerance, and diversion. However, some commonly held assumptions about these issues need to be recognized by health professionals, as outlined below.
- Opioids should be prescribed only after a thorough evaluation of the patient, consideration of alternatives, development of a treatment plan tailored to the needs of the patient and minimization of adverse effects, and ongoing monitoring and documentation.
Addiction is a risk in any patient receiving opioids, according to AAPM. Physicians must understand addiction, mitigate risk, and avoid mislabeling patients as addicts.52
Respiratory depression fears have caused withholding of opioids from patients with pain. AAPM said that risk is minimized by52
- Using extreme caution with CNS depressants, especially benzodiazepines and alcohol
- Dosing/titrating patients with sleep apnea or end-stage respiratory disease cautiously
- Knowing complete tolerance to respiratory depression risk does not develop
- Reducing opioid doses during respiratory infections or asthmatic attacks
Tolerance is diminishing analgesia with the same dose over time, AAPM indicated. Analgesic tolerance does not usually interfere with COT. Nonresponse to opioid dose escalation may reflect tolerance, disease progression, nonopioid responsive pain syndromes, or opioid-induced hyperalgesia; it requires careful evaluation.52
Diversion risk can be reduced by observing patterns of opioid prescription requests and using urine/blood drug screening, frequent follow-up, and use of PDMPs by health professionals.52
When long-term opioid therapy is to be discontinued, specific steps should be followed to minimize the effects of withdrawal. Decreases in drug dosage should be managed as follows52:
- Methadone:
- Decrease dose by 20%–50% per day until to a dose of 30 mg/day
- Then decrease by 5 mg/day every 3–5 days to 10 mg/day
- Then decrease by 2.5 mg/day every 3–5 days
- Morphine sustained release/CR:
- Decrease dose by 20%–50% per day to a dose of 45 mg/day
- Then decrease by 15 mg/day every 2–5 days
- Oxycodone CR:
- Decrease dose by 20%–50% per day to a dose of 30 mg/day
- Then decrease by 10 mg/day every 2–5 days
Some patients may have a great deal of anxiety about the potential for increased pain or onset of withdrawal symptoms. In all cases, prescribers should make decisions about tapering therapy on an individual basis.
Prescribers must also be familiar with immediate-release opioid or nonopioid augmentation, equianalgesic doses, and opioid conversion when these are used during tapering of long-term, ER/LA opioid therapy.
Pharmacists. In the pharmacy, awareness starts with knowing prescribers, their signatures, and their DEA registration numbers. Next, developing relationships with patients increases the likelihood of safe ER/LA opioid treatment. Routine assessment and dispensing measures–checking prescription dates and reviewing the patient's profile for recent fills, potential interactions, and other concerns–are also frontline measures that show patients demonstrate awareness and involvement. Pharmacists should never hesitate to ask new patients for identification and engage them in conversation to assess the situation.
Since most abusers obtain opioids from friends or relatives or by prescription from a single doctor,53 counseling at prescription handover is critical. Pharmacists need to educate patients that they must never share prescribed opioids, store them securely and dispose of them properly (discussed in more detail later in this module). The Food and Drug Administration (FDA) now requires Medication Guides for many opioids; always provide the most recent Medication Guide when any outpatient ER/LA opioid is dispensed (new or repeat prescription), and take a few minutes to review the material with patients. Doing so manages patients' expectations and offers an opportunity to clarify that these drugs are dispensed under a set of inflexible rules that cannot be violated.39,51,54
Pain contracts
A treatment contract is one method to clarify patient responsibilities and monitor adherence. Physicians and pain specialists sometimes use contracts for patients with chronic noncancer pain. For opioid analgesics, the contract explains the potential for addiction and also explains the conditions under which the physician will issue continuing prescriptions. It also indicates that the prescriber will not reissue lost or stolen prescriptions or issue prescription after hours or on weekends. Interested readers can find an example here. [http://pmp.pharmacy.state.mn.us/assets/files/PDFs/Sample Pain Management Contract.pdf]
While no evidence indicates that agreements have made any effect on decreasing opioid abuse,18 contracts formalize the prescriber–patient relationship, manage patients' expectations, and make it clear the prescriber can and will stop the opioid if the patient is irresponsible with its use.
Patient counseling
At every opportunity and patient contact, health professionals need to counsel patients and their caregivers or significant others. The FDA's Risk Evaluation and Mitigation Strategies (REMS, discussed below) require prescribers to take a thorough history from opioid patients, looking for opioid misuse/abuse risk factors of patient or family history of SUD or a patient history of psychiatric illness. They need to consider pregnancy status, current medications, and the potential that an abuser has access to legitimate user's household.39,51
Physicians, nurses, social workers, and pharmacists should discuss safe use with all opioid patients. The FDA's Medication Guide is a required element of counseling.39,51 Since patients are the leading source of opioid analgesics for abusers, appropriate storage and disposal is a critical counseling component. Opioid prescribers are required to educate patients (and possibly family members) on secure storage of their medications and appropriate disposal once the course of treatment is completed.12
FDA recommends patients dispose opioids by flushing them down the toilet, as this provides immediate, definitive elimination of safety hazards from intentional use or accidental exposure. Patients should fold transdermal patches in half before flushing.55
Systemic interventions
Systemic interventions have been developed to create roadblocks for abusers. These have been implemented by state and federal governments, insurers, regulators, and drug manufacturers.
Prescription Drug Monitoring Programs
In 2011, the DEA mandated PDMPs–automated programs that collect, analyze, and report information on CS prescribing and dispensing–in every state. Most PDMPs transfer prescription information electronically from dispensing pharmacies to a state agency. PDMPs that make real-time data available allow pharmacists and physicians to determine whether a patient is receiving CS prescriptions from multiple prescribers, and whether the patient has filled or refilled an opioid prescription.12
West Virginia's PDMP is the Controlled Substance Automated Prescription Program (CSAPP). All clinicians who prescribe or dispense opioid analgesics to patients for chronic noncancer pain must register with CSAPP, and obtain information on these patients at least annually. Prescribers and pharmacists must enter all information pertaining to CSs in Schedules II, III, or IV that are dispensed to WV residents into CSAPP within 24 hours. Before each patient search, prescribers and pharmacists must certify that they are seeking data solely for the purpose of providing health care to current patients, and that they will not allow access by unauthorized persons.56
PDMPs are available in almost every state. But underuse by pharmacists may have limited their efficacy.57
Risk Evaluation and Mitigation Strategies
ER and LA opioid analgesics are highly potent drugs, with FDA-approved indications for pain severe enough to require daily, around-the-clock, long-term opioid treatment for which alternative treatment options are inadequate.58 To ensure benefits outweigh risks, FDA enacted a classwide REMS program for proprietary and generic ER/LA opioid analgesics.39 This was followed by a REMS program for transmucosal immediate-release fentanyl (TIRF) products.54 TIRF products are available only through a restricted program called the TIRF REMS Access program. Unlike the voluntary ER/LA REMS program, enrollment is required for pharmacies, prescribers, patients, and wholesalers to purchase, dispense, prescribe, receive, or distribute TIRFs.54,59
Table 7 lists opioid analgesics covered in both REMS programs.
| Table 7. Generic and branded opioid analgesics subject to FDA REMS programsa |
| ER/LA REMS |
TIRF REMS |
|
All extended-release, oral dosage forms containing
- Hydrocodone (Zohydro, Hysingla)
- Hydromorphone (Exalgo)
- Morphine (Avinza, Embeda, Kadian, MS Contin)
- Oxycodone (OxyContin, Targiniq)
- Oxymorphone (Opana)
- Tapentadol (Nucynta)
|
Fentanyl products formulated as
- Sublingual tablets (Abstral)
- Oral transmucosal lozenges (Actiq)
- Buccal tablets (Fentora)
- Nasal spray (Lazanda)
- Buccal soluble film (Onsolis)
- Sublingual spray (Subsys)
|
| Fentanyl (Duragesic) and buprenorphine (Butrans) ER transdermal delivery systems |
| Methadone oral forms indicated for analgesic use (Methadose, Dolophine) |
| Sources: References 39, 54 |
Short-acting opioid analgesics and tramadol ER are not subject to REMS. IONSYS (fentanyl iontophoretic transdermal system) is a patient-controlled fentanyl delivery system restricted to inpatient use; it is not subject to the TIRF REMS.60
The ER/LA opioid REMS focus on prescriber education uses three approaches58:
(1) Elements to Assure Safe Use, specifically REMS-compliant, free prescriber safety education/training through accredited continuing education providers. Pharmacists are not required to complete additional education/training, verification, or enrollment to dispense ER/LA opioid analgesics.
(2) Patient Counseling Document (PCD): Contains class-wide patient safety information (Figure 1).
(3) Product-Specific Medication Guide to be dispensed by pharmacists with every new and refill prescription of products covered by the REMS.
Figure 1. Patient counseling document for extended-release/long-acting opioid analgesics.
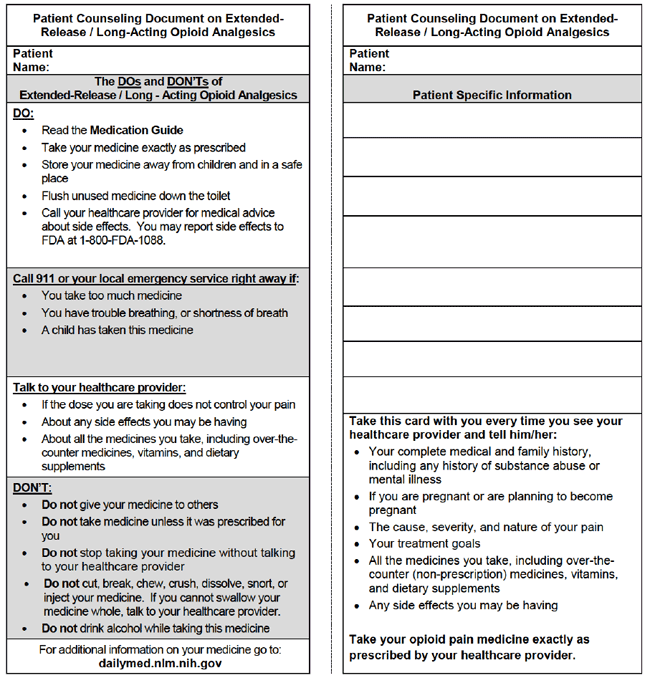 |
| Source: Daily Med, a service of the National Library of Medicine at the National Institutes of Health. https://dailymed.nlm.nih.gov/dailymed/index.cfm. |
The TIRF REMS Access program has two components54,59:
- Education program: Provides safety information essential for minimizing TIRF-associated risks, including
- Appropriate patient selection for breakthrough pain in adult cancer patients tolerant to opioid analgesic therapy for underlying persistent cancer pain. Actiq and generics approved for those age 16 years or older.
- Contraindications including opioid nontolerant patients; acute, postoperative, headache/migraine, or dental pain; intolerance/hypersensitivity to any component or to fentanyl.
- Patient-specific risk factors acknowledging accidental exposure to TIRF products can be fatal to children, people not prescribed TIRF, or those lacking opioid tolerance. Use of CYP3A4 inhibitors may elevate plasma fentanyl concentration to fatal levels.
- Dosage and administration that explicitly conveys that TIRF products lack bioequivalence, and TIRF-to-TIRF conversion on a microgram-per- microgram basis can cause fatal overdose. In each breakthrough pain episode, limit to dosage to 4 or fewer doses per day.
- Patient counseling: Requires Medication Guide (see above).
- Effective patient management and follow-up as outlined in the ER/LA REMS.
- Knowledge Assessment of TIRF products and use. Must be completed after the Education Program to prescribe or dispense TIRF products.
FDA provides online assistance regarding opioids for prescribers, patients, and pharmacists (see Table 8).
State health insurance oversight
At the state level, civil servants are evaluating state health insurance mechanisms to prevent prescription drug misuse, diversion, and overdose (e.g., via Medicaid or workers' compensation). Legislators are evaluating state-level laws, policies, and regulations (e.g., legislation regulating pain clinics, or supporting clinical practice guidelines).
WV Medicaid limits the number of units or doses that can be prescribed for opioid pain relievers and benzodiazepines. To prescribe a larger quantity, a prescriber must submit a clinical justification and request a waiver. WV Medicaid's active Lock-In program scans prescription records monthly to look for inappropriate use of controlled substances. Medicaid beneficiaries who meet or exceed the program's criteria are locked into a single pharmacy that monitors the beneficiary's controlled substance use.61
WV's worker's compensation program requires prescribers to navigate a step process to prescribe opioids to injured workers. It employs the Presley Reed Guide, workplace
guidelines for expected duration of treatment, and physicians must document patients' substantial reduction in pain intensity and continuing improvement to continue prescribing opioids.62
Legislation
In addition to laws discussed elsewhere in this review, WV is taking steps to address opioid abuse and deaths. Since May 2015, the Access to Opioid Antagonists Act has allowed police officers, first responders, as well as family and friends of opioid users to carry and administer the opioid overdose antidote naloxone.63 Pharmacists should be familiar with the naloxone products available without a prescription and how to administer them (discussed below). In addition, overdose is a medical emergency; if naloxone is needed, an emergency plan should be activated including a 911 call.
Abuse-deterrent formulations
A final systemic intervention is being implemented at the drug development level. Abuse-deterrent formulations (ADFs) are products with built-in physical- or chemical- tampering barriers. Of currently approved ADFs, Embeda (morphine ER) and Targiniq (oxycodone ER) contain inert naltrexone – activated and released only through tampering – to reduce or defeat euphoria and reward. All other ADFs use tamper- resistance features intended to prevent chewing, crushing for intranasal use, or dissolving for intravenous use.30,47
ADF OxyContin was introduced in 2010 to curtail wide-scale OxyContin abuse. Abuse rates fell quickly after its introduction. The decline resulted from migration to other opioids, primarily heroin. By 2012, abusers had adapted, a ceiling effect emerged, and no further decreases occurred. Some users switched to nonoral routes or other drugs. Approximately one-third of OxyContin abusers figured out how to defeat the ADF mechanism and continued to inhale or inject this product. Some simply continued to use the new formulation orally.23
ADFs can reduce abuse prevalence of the reformulated opioid, but significant levels of residual abuse may persist.23 Manufacturers are searching for better approaches. These include using prodrugs and intranasal, transdermal, and depot delivery systems. The challenge is great; new formulations must deter abuse but remain effective, avoid introducing new safety issues for patients with legitimate needs, and ensure safety. Ideally, new ADFs would meet all these challenges and be cost-effective.46
Naloxone
Naloxone is now the standard of care for opioid overdose, and clinicians should recommend it for all patients exposed to opioids regardless of the source. In community settings, it is generally administered intranasally, intramuscularly, or subcutaneously.64-66 Naloxone is a bystander-administered drug.
This opioid antagonist is a synthetic oxymorphone congener created by replacing oxymorphone's methyl group on the nitrogen atom with an allyl group. Although molecularly similar to opioids, it is a pure opioid antagonist. It has no agonist or opioid- like properties and provides no analgesia, dysphoria, or pharmacologic activity other than opioid reversal.64-66 It appears that naloxone antagonizes opioid effects by competing for CNS mu, kappa, and sigma opiate receptor sites, with greatest affinity for the mu receptor.
Naloxone reverses opioids' effects (e.g., analgesia, respiratory depression, sedation, hypotension, and dysphoria).64 Additionally, naloxone can reverse the psychotomimetic and dysphoric effects of agonist-antagonists (i.e., pentazocine or buprenorphine). When administered to patients who are dependent on opioids, naloxone produces withdrawal symptoms (flushing, dizziness, tiredness, weakness, nervousness, restlessness, irritability, body aches, diarrhea, stomach pain, nausea, fever, chills, piloerection [goosebumps], sneezing, shortness of breath, or runny nose) within minutes; these symptoms usually subside over approximately 2 hours. The severity and duration of withdrawal depends on the naloxone dose and the type and severity of the patient's opioid dependence. As naloxone's effects wear off, patients may slip back into sedation depending on the duration of action of the opiate on which they overdosed.64,-66
All naloxone formulations lack adverse effects (AEs); given to anyone who has not taken an opiate, it produces no effects. Administered to an individual who has taken opioids, withdrawal symptoms are often mistaken for AE. These symptoms are not life- threatening. Because of the short half-life of naloxone, the withdrawal symptoms are also short-lived.64-67
Serious withdrawal concerns include hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events.64-67
Naloxone for outpatient or home use is available in several different forms. Manufacturers have created formulations that can be administered by intramuscular (IM), subcutaneous, intranasal (IN), and intravenous (IV) routes.64-66 Each product comes with ample information describing its use, which pharmacists must review with the consumer. The counseling session should cover these key points68:
- Bystanders should administer the naloxone, then call 911. They should then administer rescue breathing to prevent organ damage.
- Once bystanders call 911, ambulance dispatchers will usually coach them until help arrives.
- Naloxone works in 2 to 5 minutes depending on its method of administration; take-home methods generally work in 5 minutes; naloxone's effects last 30 to 60 minutes.
- The bystander must stay with the overdose victim until help arrives; additional doses of naloxone may be needed.
- Naloxone will not reverse overdoses from nonopioid drugs and will not harm someone if they are not taking opioids and it is given by mistake.
- Naloxone is safe for pregnant women.
CASE STUDY
A case study illustrates several important elements of best practices in pharmacists' care of patients receiving opioid therapy for noncancer pain. These key points are italicized as the case is described in this section.
Wally Smith is a new patient to your pharmacy. He appears with a prescription for OxyContin 80 mg. You ask him if he has taken OxyContin before, and he tells you he is a laborer who has been on worker's compensation for several months. He is being treated for back pain, and had been having his prescriptions filled across town at a pharmacy well known to you. He recently moved and plans to use your pharmacy because it is closer to his new home.
At this point, Mr. Smith seems forthcoming and there is no indication that he is anything other than a patient with a legitimate need for this analgesic. You access CSAPP and examine Mr. Smith's history, and spend some time talking to Mr. Smith to get a sense of his history and character–these are best practices.
In your discussion with Mr. Smith, you learn that he is 24-years-old, and had worked for his company for 3 years when he had the accident that led to his pain problem. He says he would like to return to work, and asks if you would have time to talk to him about how to manage his constipation. He is a smoker (you can smell it on his clothing), and he says he doesn't drink since he started taking opioid analgesics. You identify smoking and chronic pain as risk factors for OUD.
For 3 months, Mr. Smith returns at appropriate times with prescriptions for OxyContin 80 mg daily. At the start of the 4th month, Mr. Smith's physician begins to taper the drug. Mr. Smith says that his pain is controlled and he wants to return to works as soon as possible. He presents a prescription for "OxyContin 30 mg daily–please return to clinic in 28 days." At this point, the appropriate action is for you, as the dispensing pharmacist, to call the prescriber and question the dose; the doctor has reduced the dose too quickly and the patient will need to be seen sooner than 28 days from now.
Mr. Smith's physician responds positively to a more gradual taper of the opioid therapy. When the opioid dose reaches 30 mg daily, Mr. Smith asks the physician to increase the dose because the back pain has recurred after several days at that dose. The physician asks Mr. Smith to try exercise, meditation, hot and cold packs, and physical therapy. He does not taper any further. After 3 weeks at the 30-mg dose. Mr. Smith visits the local emergency departments one evening and later presents a prescription at the pharmacy for hydrocodone for breakthrough pain. He says he will tell the doctor about this prescription when he sees him next week. Although addiction may be a possibility, it is also possible that Mr. Smith is exhibiting pseudo-addiction because his pain is unrelieved.
Mr. Smith's prescriber increases his OxyContin dose to 40 mg daily, and Mr. Smith continues to fill prescriptions appropriately. Worker's compensation continues to pay for his medication. A few months later, Mr. Smith presents a prescription for OxyContin 40 mg daily, #28, written by a new physician; Mr. Smith says he will pay cash for the prescription. When you ask him if he is still seeing his regular pain specialist, he says yes. When you ask him why he has this prescription, he mumbles something about losing his medication. Faced with this new turn of events, you should not fill the prescription and allow Mr. Smith to pay cash –this would be inappropriate. Instead, you should tell Mr. Smith you need to ask his regular prescriber whether you should fill this prescription. Collaboration between prescribers is needed to make sure everyone knows what Mr. Smith's situation is.
Mr. Smith appears at the pharmacy 2 weeks later and has a prescription from his regular provider. He appears a few minutes before closing, and he is unkempt and smells of alcohol. Although the prescriber's signature looks genuine, the prescription has no DEA number on it and indicates that Mr. Smith can have two refills. You tell Mr. Smith that you need to call the prescriber in the morning, and he can pick up the prescription tomorrow afternoon. Mr. Smith becomes surly and demands to have the prescription now. The best action it to remind Mr. Smith that, as you have explained carefully and repeatedly before, the rules about controlled substances allow no room for creativity and he will have to wait until tomorrow. Good counseling and provision of the product's Medication Guide at every encounter manages patient's expectations.
Shortly after this encounter, you learn that the provider had forgotten to put his DEA number on the prescription, but Mr. Smith had added the refills. Mr. Smith's provider has a rule that after a patient exhibits drug-seeking behavior, he must sign a treatment contract. Mr. Smith agrees to sign the contract and the provider faxes a copy to you for the pharmacy files. The contract informs patients that they cannot get prescriptions on nights or weekends.
Within a few months, Mr. Smith violates the terms of his contract and the prescriber notifies him that he will no longer treat him. Worker's compensation terminates his claim. From time to time, Mr. Smith presents prescriptions for opioids. Sometimes these are for OxyContin, sometimes MS-Contin, and sometimes a short-acting opioid. You suspect that he is buying opioids in the street, as CSAPP shows only intermittent prescriptions.
Several month later, a regular customer, Mrs. White, presents a prescription for Evzio (naloxone hydrochloride injection) Auto-Injector. This surprises you because she is pregnant, clearly healthy, and not an opioid abuser. She explains that she is Mr. Smith's sister and he lives with her. He has had several close calls lately, and landed in the emergency department. The emergency department physician suggested she carry a naloxone auto-injector. WV allows people who are close to people who have drug abuse problems to carry naloxone. Since May 2015, the Access to Opioid Antagonists Act allows police officers, first responders, and family and friends to carry and administer naloxone.
Mr. Smith finally acknowledges that he has an opioid addiction and is accepted into a treatment program. Before he goes, he and his sister come to the pharmacy and ask you to explain what they can expect in terms of drug treatment. You tell them that treatment programs usually tailor their approach to individual patients' needs. You tell him that most treatment facilities use medically supervised detoxification followed by maintenance with opioid substitution therapies. Patients value the advice and counsel of a trusted pharmacist with whom they have a long-term professional relationship.
Mr. Smith completes 4 months of inpatient treatment for his drug addiction. He is discharged on buprenorphine 16 mg/naltrexone 4 mg combination sublingual filmtabs. Unemployed and uninsured, he is finding the cost to be a burden. He asks you if he will ever be able to get off this medication or if there is another, less expensive medication available. Cost is problem for many patients. You can say that some patients with opioid use disorders can taper their substitution therapy gradually and transition to naltrexone. He will need to work with his program to see if this is an option for him.
CONCLUSION
The sharply increasing rates in opioid analgesic nonmedical use, overdose deaths, and treatment-seeking during 2002–11 have been followed by declining or stable rates nationwide and in West Virginia during subsequent years. Accounting for this trend is abuser migration from opioid analgesics to heroin following introduction of tamper- resistant opioid analgesic formulations, and greater use of state PDMPs. More difficult to measure is impact from other recently implemented risk reduction strategies.
Given changes in the proportion of ED visits and fatal overdoses involving patients prescribed opioid analgesics, it is likely that changes in opioid conversion methods, higher awareness of secure storage and safe disposal, and prescriber and pharmacist involvement in FDA opioid analgesic REMS programs are reducing risks associated with
opioid analgesic prescribing, while ensuring their access by pain patients in need. Federal efforts to reduce opioid analgesic-related mortality have emphasized patient monitoring, securing controlled substance supplies, and educating prescribers and patients through FDA REMS programs. Other risk reduction strategies–heightened awareness of the problem, increased education at the state level, use of pain guidelines, interdisciplinary collaboration, and improved opioid conversion methods–are key parts of the necessary multifaceted response.
REFERENCES
- Chen LH, Hedegaard H, Warner M. Drug-poisoning Deaths Involving Opioid Analgesics: United States, 1999–2011. NCHS data brief, no 166. Hyattsville, MD: National Center for Health Statistics. 2014.
- Centers for Disease Control and Prevention. Opioid overdose: U.S. opioid prescribing rate maps. Accessed at https://www.cdc.gov/drugoverdose/maps/rxrate-maps.html, February 20, 2019.
- Wilson R. Drug overdoses kill more people than auto accidents in 29 states. Washington Post. October 8, 2013. Accessed at http://www.washingtonpost.com/blogs/govbeat/wp/2013/10/08/drug-overdoses-kill- more-people-than-auto-accidents-in-29-states/, November 1, 2016.
- Smith J. West Virginia ranks first in drug overdose deaths. Accessed at http://wvmetronews.com/2013/10/08/west-virginia-ranks-first-in-drug-overdose-deaths/, November 1, 2016.
- Frenk SM, Porter KS, Paulozzi LJ. Prescription opioid analgesic use among adults: United States, 1999–2012. NCHS data brief no. 189; 2015. Accessed at http://www.cdc.gov/nchs/data/databriefs/db189.htm, November 1, 2016.
- Substance Abuse and Mental Health Services Administration (SAMHSA). National Survey on Drug Use and Health: Comparison of 2002-2003 and 2012-2013 Model-Based Prevalence Estimates (50 States and the District of Columbia). February 10, 2015. Accessed at http://www.samhsa.gov/data/sites/default/files/NSDUHsaeLongTermCHG2013/NSDUH saeLongTermCHG2013.pdf, November 1, 2016.
- CDCR. Rates of deaths from drug poisoning and drug poisoning involving opioid analgesics – United States, 1999–2013. MMWR Morb Mortal Wkly Rep. 2015;64:32.
- Jones CM, Paulozzi LJ, Mack KA; Centers for Disease Control and Prevention (CDC). Alcohol involvement in opioid pain reliever and benzodiazepine drug abuse-related emergency department visits and drug-related deaths – United States, 2010. MMWR Morb Mortal Wkly Rep. 2014;63(40):881-5.
- WVDFWF 2012. West Virginia Governor's Drug-Free Work Force. What you should know about drug abuse in West Virginia. 2012. Access at http://www.faceyourfuturewv.com/real-drug-facts-side.html, November 1, 2016.
- Warner M, Hedegaard H, Chen L-H. Trends in drug-poisoning deaths involving opioid analgesics and heroin: United States, 1999–2012. Health E-Stats. December 2014.
- Prince V. Pain management in patients with substance-use disorders. In: Chronic Illnesses I, II, and III–PSAP-VII, Book 5. American College of Clinical Pharmacy, 2011. Accessed at www.accp.com/docs/bookstore/psap/p7b05.sample03.pdf. Accessed November 1, 2016.
- Federation of State Medical Boards (FSMB). Model Policy for the Use of Opioid Analgesics in the Treatment of Chronic Pain. Washington, DC: The Federation, 2013. Accessed at https://www.fsmb.org/siteassets/advocacy/policies/opioid_guidelines_as_adopted_april-2017_final.pdf, February 22, 2019.
- Utah Department of Health. Utah Clinical Guidelines on Prescribing Opioids for Treatment of Pain. Salt Lake City, UT: Utah Department of Health. 2009. Accessed at http://health.utah.gov/prescription/pdf/guidelines/final.04.09opioidGuidlines.pdf, November 1, 2016.
- American Psychiatric Association. Changes from DSM-IV-TR to DSM-5. 2013. Accessed at http://www.dsm5.org/Documents/changes from dsm-iv- tr to dsm-5.pdf. Accessed November 1, 2016.
- American Society of Addiction Medicine (ASAM). Definition of Addiction. Last updated 2011. Accessed at https://www.asam.org/resources/definition-of-addiction, November 1, 2016.
- Tormoehlen LM, Mowry JB, Bodle JD, Rusyniak DE. Increased adolescent opioid use and complications reported to a poison control center following the 2000 JCAHO pain initiative. Clin Toxicol (Phila). 2011;49(6):492–498.
- Anonymous. 2001 to 2010: The decade of pain control and research. Accessed at http://lifeinpain.org/node/141. Accessed November 1, 2016.
- Schatman ME, Webster LR. The health insurance industry: perpetuating the opioid crisis through policies of cost-containment and profitability. J Pain Res. 2015:8:153–158.
- Medical Treatment Utilization Schedule (MTUS). Chronic Pain Medical Treatment Guidelines. State of California, Department of Industrial Relations, Division of Workers' Compensation (DWC). 2009. Accessed at http://www.dir.ca.gov/dwc/DWCPropRegs/MTUS_Regulations/MTUS_ChronicPainMed icalTreatmentGuidelines.pdf, November 1, 2016.
- Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: controversies, current status, and future directions. Exp Clin Psychopharmacol. 2008;16(5):405–416.
- Buckenmaier CC, Gallagher RM, Cahaba A, et al. War on pain. New strategies in pain management for military personnel and veterans. Fed Pract Suppl. 2011 June:1-16.
- King NB, Fraser V, Boikos C, et al. Determinants of increased opioid-related mortality in the United States and Canada, 1990–2013: a systematic review. Am J Public Health. 2014;104:e32–e42.
- Cicero TJ, Ellis MS. Abuse-deterrent formulations and the prescription opioid abuse epidemic in the United States: lessons learned from OxyContin. JAMA Psychiatry. 2015;72(5):424-430.
- Cicero TJ, Ellis MS, Surratt HL, Kurtz SP. Factors influencing the selection of hydrocodone and oxycodone as primary opioids in substance abusers seeking treatment in the United States. Pain. 2013;154(12):2639-48.
- Substance Abuse and Mental Health Services Administration (SAMHSA), Center for Behavioral Health Statistics and Quality. Treatment Episode Data Set (TEDS): 2001- 2011. State Admissions to Substance Abuse Treatment Services. Rockville, MD: Substance Abuse and Mental Health Services Administration, June 2013. Accessed at http://www.samhsa.gov/data/sites/default/files/TEDS2011St_Web/TEDS2011St_Web/T EDS2011St_Web.pdf, November 1, 2016.
- Office of National Drug Control Policy. West Virginia Drug Control Update. Accessed at https://obamawhitehouse.archives.gov/sites/default/files/docs/state_profile_-_west_virginia_0.pdf, November 1, 2016.
- Moseley GL. Reconceptualising pain according to modern pain science. Phys Ther Rev. 2007;12:169–178.
- Drug Enforcement Administration. Pharmacist's Manual. United States Department of Justice. Revised 2010. Accessed at http://www.deadiversion.usdoj.gov/pubs/manuals/pharm2/index.html, November 1, 2016.
- Cicero TJ, Kurtz SP, Surratt HL, et al. Multiple determinants of specific modes of prescription opioid diversion. J Drug Issues. 2011;41(2):283-304.
- Tufts Health Care Institute. Perspectives on Opioids for Pain Management: Optimizing the Benefits, while Minimizing the Risks. Tufts Health Care Institute Program on Opioid Risk Management, 2013. Accessed at http://www.thci.org/opioid/, November 1, 2016.
- Webster LR, Fine PG. Overdose deaths demand a new paradigm for opioid rotation. Pain Med. 2012;13:571–574.
- Hoppe J, Howland MA, Nelson L. The role of pharmacies and pharmacists in managing controlled substance dispensing. Pain Med. 2014(Dec);15(12):1996-8.
- Wallace LS, Keenum AJ, AbdurRaqeeb O, et al. Terminology matters: patient understanding of "opioids" and "narcotics." Pain Pract. 2013;13:104–108.
- American Academy of Family Physicians. Under-treatment of pain: resources for pain management. 2009. Accessed at http://www.aafp.org/patient-care/public- health/pain-opioids.html, November 1, 2016.
- Walwyn WM, Miotto KA, Evans CJ. Opioid pharmaceuticals and addiction: the issues, and research directions seeking solutions. Drug Alcohol Depend. 2010;108(3):156–165.
- Bieber CM, Fernandez K, Borsook D, et al. Retrospective accounts of initial subjective effects of opioids in patients treated for pain who do or do not develop opioid addiction: a pilot case-control study. Exp Clin Psychopharmacol. 2008;16(5):429–434.
- Oliver J, et al.; the International Nursing Society on Addiction and the American Society for Pain Management Nursing. American Society for Pain Management Nursing Position Statement: pain management in patients with substance use disorders. Pain Manage Nurs. 2012;13:169-183.
- McPherson ML. Opioids: fears, myths, and misconceptions. PainView. 2011;7:16-18.
- Food and Drug Administration. Extended-Release (ER) And Long-Acting (LA) Opioid Analgesics Risk Evaluation And Mitigation Strategy (REMS). Most Recent Modification: 06/2015. Accessed at http://www.accessdata.fda.gov/drugsatfda_docs/rems/ERLA_opioids_2015.06.26_REMS _document.pdf, November 1, 2016.
- VA/DoD. Clinical practice guidelines. Management of opioid therapy (OT) for chronic pain. 2017. Accessed at https://www.healthquality.va.gov/guidelines/pain/cot, February 20, 2019.
- Peirce GL, Smith MJ, Abate MA, Halverson J. Doctor and pharmacy shopping for controlled substances. Med Care. 2012;50(6):494–500.
- Savage SR, Kirsh KL, Passik SD. Challenges in using opioids to treat pain in persons with substance use disorders. Addiction Sci Clin Pract. 2008;4(2):4-25.
- Drug Enforcement Administration. Recognizing the drug abuser. Accessed at https://www.deadiversion.usdoj.gov/pubs/brochures/pdfs/recognizing_drug_abuser_trifold.pdf, November 1, 2016.
- Sloan PA, Davis MP, Gamier P. ER/LA opioid REMS supplement. J Opioid Manage. 2014;10:1-48.
- Jones CM, Paulozzi LJ, Mack KA. Sources of prescription opioid pain relievers by frequency of past-year nonmedical use United States, 2008-2011. JAMA Intern Med. 2014;174(5):802-803.
- Wick JY. Drug-abuse deterrent formulations. Consult Pharm. 2009;24(5):356-62, 365.
- Simon K, Worthy SL, Barnes MC, Tarbell B. Abuse-deterrent formulations: transitioning the pharmaceutical market to improve public health and safety. Ther Adv Drug Saf. 2015;6:67–79.
- Cicero TJ, Ellis MS, Paradis A, Ortbal Z. Determinants of fentanyl and other potent µ opioid agonist misuse in opioid-dependent individuals. Pharmacoepidemiol Drug Saf. 2010;19:1057-63.
- Wightman R, Perrone J, Portelli I, Nelson L. Likeability and abuse liability of commonly prescribed opioids. J Med Toxicol. 2012;8:335–340.
- Atkinson TJ, Fudin J, Pandula A, et al. Medication pain management in the elderly: unique and underutilized analgesic treatment options. Clin Ther. 2013;35:1669-1689.
- ER/LA Opioid Analgesics REMS frequently asked questions. The ER/LA opioid analgesics REMS program companies (RPCs). Last updated July 8, 2015. Accessed at http://www.er-la-opioidrems.com/IwgUI/rems/faq.action#pharmacist, November 1, 2016.
- American Academy of Pain Medicine. Use of opioids for the treatment of chronic pain. February 2013. Accessed at https://painmed.org/clinician-resources/tools-for-practice, November 1, 2016.
- Substance Abuse and Mental Health Services Administration, Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings, NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2014. Accessed at http://www.samhsa.gov/data/sites/default/files/NSDUHresultsPDFWHTML2013/Web/N SDUHresults2013.pdf, November 1, 2016.
- TIRF REMS Access Program. Education Program for Prescribers and Pharmacists. Accessed at https://www.tirfremsaccess.com/TirfUI/rems/pdf/education-and-ka.pdf, November 1, 2016.
- FDA Consumer Health Information. Where and How to Dispose of Unused Medicines. U.S. Food and Drug Administration, October 2017. Accessed at https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm101653.htm, February 20, 2019.
- West Virginia Board of Pharmacy. Controlled substance monitoring program. Accessed at https://www.csappwv.com, November 1, 2016.
- Fleming ML, Chandwani H, Barner JC, Weber SN, Okoro TT. Prescribers and pharmacists requests for prescription monitoring program (PMP) data: does PMP structure matter? J Pain Palliat Care Pharmacother. 2013;27:136–142.
- McNaughton EC, Black RA, Weber SE, Butler SF. Assessing abuse potential of new analgesic medications following market release: An evaluation of Internet discussion of tapentadol abuse. Pain Med. 2015;16:131–140.
- TIRF REMS Access: Frequently Asked Questions (FAQs). Accessed at https://www.tirfremsaccess.com/TirfUI/rems/pdf/faq.pdf#page=9, November 1, 2016.
- The Medicines Company. Ionsys (fentanyl iontophoretic transdermal system) REMS. Last update 4/30/2015. Accessed at http://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforP atientsandProviders/UCM446439.pdf, November 1, 2016.
- West Virginia Department of Health & Human Services. West Virginia Medicaid is a guardian against opioid pain reliever and benzodiazepine abuse. DUR Capsules. March 2015. Accessed at https://dhhr.wv.gov/bms/BMS%20Pharmacy/Documents/DUR/Dur%20Newsletters/WV%20Newsletter%20March%202015.pdf, November 1, 2016.
- Anonymous. State opioid rules by restriction. Signature Series, October 2015. Accessed at http://www.progressive-medical.com/LinkClick.aspx?fileticket=6- eacnSITIU%3d&tabid=388, November 1, 2016.
- Wright J, Marra A. Concerns raised over W.Va. naloxone bill. WV Public Broadcasting. May 25, 2015. Accessed at http://wvpublic.org/post/concerns-raised-about- wva-naloxone-bill, November 1, 2016.
- Evzio (naloxone hydrochloride injection) [prescribing information]. Richmond, VA: kaleo, Inc; April 2014
- Narcan (naloxone hydrochloride) nasal spray [prescribing information]. Radnor, PA: Adapt Pharma, Inc.; November 2015.
- Naloxone hydrochloride injection [prescribing information]. Lake Forest, IL: Hospira, Inc.; January 2007.
- Kelly A, Kerr D, Dietze P, Patrick I, Walker T, Koutsogiannis Z. Randomised trial of intranasal versus intramuscular naloxone in prehospital treatment for suspected opioid overdose. Med J Aust. 2005;182(1):24-27.
- College of Psychiatric & Neurologic Pharmacists. Naloxone access: a practical guide for pharmacists. CPNP website. Accessed at https://cpnp.org/guideline/naloxone, November 22, 2016.
Back to Top