
Expired activity
Please go to the PowerPak
homepage and select a course.
CARING FOR COLD SORES IN THE COMMUNITY PHARMACY
INTRODUCTION
Cold sores — lesions on or near the lips — are a common complaint among consumers seeking over-the-counter (OTC) remedies in pharmacies. Known scientifically as herpes simplex labialis (HSL) and colloquially as “fever blisters,” cold sores are caused by the herpes simplex virus (HSV), which is ubiquitous in people around the world. An estimated 70% to 90% of adults have antibodies to the type 1 form of HSV (HSV1), the most common cause of cold sores.1
To provide contemporary care to patients with cold sores, community pharmacists need to know about the etiology and epidemiology of HSL and currently available OTC and prescription therapies that can affect the disease course and/or reduce symptoms of pain and tingling and the discomfort of weeping and crusting lesions in later phases of the disease.
ETIOLOGY AND CLINICAL COURSE OF HSV INFECTIONS
According to the World Health Organization (WHO), an estimated 3.7 billion people under the age of 50 years are infected by HSV1.2 Once the virus is present, it remains in the body for life. Most of the time, the virus is in a latent form that produces no symptoms. Periodic outbreaks occur in some, but not all, of those infected; many people have the virus but do not know it.
Transmission of HSV1 is usually through direct contact of a recipient’s mucosal membranes or abraded skin with mucosal secretions, saliva, or lesions of an infected patient.3 Even without symptoms or the presence of cold sores, infected patients continue shedding virus and infecting those with whom they come into contact. While HSV can infect animals, reservoirs or vectors involving nonhumans are not involved in transmission to people.1
Once primarily transmitted through genital contact or by maternal transfer to newborns during delivery, the type 2 strain of HSV is now more frequently identified as a cause of infection in the oral region. Both HSV1 and HSV2 can cause oral and genital symptoms, as well as a number of other conditions such as gingivostomatitis, keratoconjunctivitis, encephalitis, and neonatal herpes. Once present, these viruses remain in latently infected ganglia in a nonreplicating state for the lifetime of the host (Figure 1).1
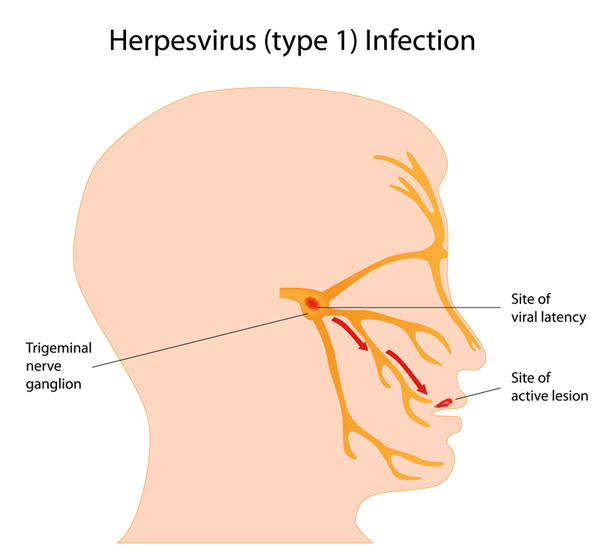
Figure 1. Sites of herpes simplex virus latency and active lesions
Risk factors for HSL include female gender, older age (65–74 years), white race, frequent upper respiratory infections, low lymphocyte counts, and lower socioeconomic status. Infants and young children with atopic dermatitis (eczema) and other allergic conditions are particularly susceptible to acquiring HSV; adults and older children with known HSV-positive status should avoid close contact with susceptible young children whenever possible. Patients with eczema who have HSL or genital herpes outbreaks during a flare can develop a serious HSV condition, eczema herpeticum. Others at risk for complications of HSV and HSL include people living with human immunodeficiency virus (HIV), patients with cancer or severe burns, and those undergoing cancer chemotherapy and antirejection treatments following transplant. Complications can include spread of HSV to the fingertips (herpes whitlow), eyes, or other organs, including the spinal cord and brain.4,5
The first time HSV1 occurs, most patients are asymptomatic, but a few experience fever, sore throat, headache, muscle aches, painful gums, and swollen lymph nodes.4 Infected persons then periodically experience outbreaks. These can be triggered when the latent virus is reactivated by stimuli such as axonal injury, fever, physical or emotional stress, menstruation, or exposure to ultraviolet light.
Five stages of a recurrent cold sore episode are evident, usually lasting for up to 2 weeks: day 1, prodrome symptoms such as tingling, burning, or itching around the mouth; day 2 or 3: blister stage, an outbreak of fluid-filled blisters along the border where the lips meet the skin of the face (Figure 2); day 4, ulcer or weeping stage (most contagious stage), blisters merge, rupture, and ooze fluid and leave shallow, reddish, open sores; days 5 to 8, crusting stage, blisters dry up and form a yellowish or brownish crust that eventually falls off; and days 9 to 12, healing stage, series of scabs form on the lesion, each smaller than the last, generally ending with complete healing (no scars remain).4,6,7
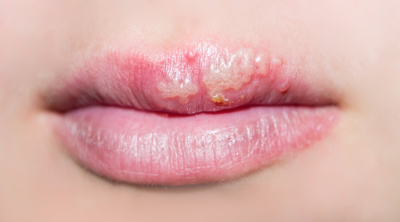
Figure 2. Herpes simplex virus lesions on the lips
As each cold sore episode begins, the virus travels down the axon to the skin or mucous membrane where replication begins and visible lesions develop. Immunocompromised patients are at higher risk of developing severe HSV infections.1,5 In immunocompetent patients, diagnosis is generally by history and presenting symptoms. Immunocompromised patients, especially those with atypical HSL symptoms, may require laboratory confirmation of the causative organism.5
Educating patients about HSV transmission is important, particularly with regard to transmission during orogenital sexual contact regardless whether lesions are present (see “Role of the Pharmacist” section for counseling points). Young adults who are at increased risk of transmission have been identified as appropriate targets of mass-education campaigns. Pregnant women at risk for or with genital herpes also need information about lowering the risk of transmission to newborns during delivery.8
Cold sores are different from canker sores. Cold sores are more common outside the mouth, while canker sores occur inside the mouth. Canker sores are not infectious; they are caused by tissue injury from a tooth, dental appliance, or acidic foods; stress; or, in complex cases, a nutritional deficiency or health condition such as Crohn’s disease or celiac disease. The pharmacist must differentiate between the 2 types of oral lesions before recommending referral or therapy.
COMMON OTC TREATMENTS FOR COLD SORES
Pharmacists are important sources of information for patients at risk for HSL outbreaks. In a prescribing study conducted in the Canadian province of Saskatchewan, cold sores were the most common minor ailment for which patients sought care (34.4% of 125 participants).9 Benefits of allowing pharmacists to control availability of oral antiviral products for this condition were reviewed in an article from New Zealand; these included cost savings to patients, permitting patients access to products more quickly, and initiating treatment earlier in the prodome of an HSL outbreak.5
In the United States (U.S.), several categories of OTC agents are available for the management of HSL. These include sunscreens, emollients and moisturizers, topical analgesics/anesthetics, and antiviral agents. Examples of these topical agents and products are listed in Table 1.3–7 While evidence supporting the efficacy for many agents is weak or nonexistent, OTC products are generally considered useful for decreasing the symptoms of HSL during an outbreak, especially tingling, irritation, and pain.
| Table 1. Over-the-Counter Options for the Topical Management of Herpes Labialis3–7 |
| Mechanism of action |
Examples of products (brand names) |
Application/populations
approved for use |
Comments |
| Analgesics/antiseptics |
Camphorated phenol (Campho-Phenique, store brands) |
Adults and children ≥2 years old
Apply 1 to 3 times daily |
Do not cover with a bandage |
| Anesthetics |
Lidocaine (Zilactin-L)
Benzocaine (Kank-a, Anbesol, Zilactin-B)
Benzyl alcohol (Zilactin) |
Adults and children ≥2 years old
Apply up to 4 times daily |
Contraindicated in patients with hypersensitivity to local anesthetics in this pharmacologic class
Do not get product in eyes
Refer to product package insert for active and inactive ingredients |
| Antivirals |
Docosanol 10% (Abreva) |
Adults and children ≥12 years old
Apply 5 times daily until healed |
Use at first symptom of a cold sore and continue therapy through healing of lesions
Discontinue use if allergies develop; severe reaction possible |
| Moisturizers |
Lip balms (e.g., Vaseline, Aquaphor); some contain sunscreens |
Population depends on product(s) selected
Apply as often as needed |
Review product active and inactive ingredients |
| Sunscreens |
Dimethicone/sunscreens (Herpecin-L) |
Adults and children ≥12 years old
Apply as often as needed; at least every 2 hours when in the sun |
General sunscreen products applied to exposed skin on the face can be used on a long-term basis to protect against both herpes labialis outbreaks (in susceptible patients) and other dermatologic conditions, including skin cancer |
Sunscreens
Exposure to sunlight or ultraviolet light has been identified as a trigger for HSL in some patients. Therefore, sunscreens and ultraviolet-rated protectants on the lips are reasonable preventive measures for patients who develop cold sores several times each year or during periods of sun exposure.10 The degree of ultraviolet protection afforded by some lip-protectant products is minimal (e.g., sun protection factor [SPF] ratings of 15); the American Academy of Dermatology (AAD) recommends an SPF of at least 30 for optimizing cold sore prevention.7 In a recent meta-analysis, evidence showed that sunscreen prevented cold sores induced by ultraviolet light but not by natural sunlight.11 Long-term use of sunscreens and ultraviolet-protectant lip balms is a reasonable recommendation to make for patients with recurrent HSL.
Lip balms and protectants
Lip balms can be used to keep lesions covered to decrease discomfort as they erupt, crust over, and heal. Lip balm and topical ingredients, both active and inactive, are important to review with patients, as some ingredients are beneficial but others may worsen the cold sore through irritant or patient-specific hypersensitivity actions. Ingredients such as white petrolatum, allantoin, calamine, oxide, and cocoa butter usually help relieve dryness and cracking. These are appropriate for use in topical application on the cold sore/lip area, particularly in later stages of the clinical course.12
Propolis (bee’s wax) is an agent that has become increasingly popular as people have used it for many different conditions; however, evidence to support its use is conflicting. Although a small study showed that propolis can reduce the duration and pain of cold sores,13 other literature has shown that propolis is a possible irritant to the skin and should not be recommended.14 Patients who are allergic to bees or have asthma should avoid propolis products.
Several ingredients in lip balm or topical products can worsen the cold sore lesion or cause additional adverse dermatologic effects. Ingredients such as vitamin E, ricinoleic acid (castor oil), balsam of Peru, lanolin, and fragances have been shown to potentially irritate the cold sore and/or result in dermatitis- like reactions.14 Additionally, Pray has recommended against use of products marketed for cold sores that contain salicylic acid, even as an inactive ingredient (e.g., Carmex), because of its keratolytic properties.12 Salicylic acid acts by eroding thickened skin surfaces such as warts and calluses, and, when applied on the cold sore, where the skin is not as thick, it could cause further skin damage. Overall, products that contain any of these potentially irritating ingredients should be discussed with the patient to enhance selection and use of OTC topical products.
Anesthetics and analgesics
Topical products containing local anesthetics in the ester and amide (“–caine”) classes are commonly used to decrease discomfort once lesions appear, erupt, and crust over (Table 1).3–7 These products require frequent, daily applications to achieve optimal outcomes in pain reduction and healing. In a double-blind, placebo-controlled trial of 72 patients, tetracaine administered topically as a 1.8% cream showed a significant reduction in healing time of recurrent HSL lesions, but the drug has not been developed commercially for this use.15,16
For patients with pain not relieved by topical therapy, oral OTC analgesics can be recommended. Ibuprofen or acetaminophen in standard OTC doses is generally effective.17
OTC antiviral agent
The only OTC product approved by the U.S. Food and Drug Administration (FDA) for decreasing the duration of HSL outbreaks is docosanol, a saturated 22-carbon aliphatic alcohol (Table 1). 3–7 This antiviral agent acts through a unique mechanism of action, one that is unlikely to produce viral resistance. While most antiviral compounds useful for herpetic infections are nucleoside analogues or related agents, docosanol reduces the duration of HSL symptoms by inhibiting the fusion of the host cell plasma membrane and the HSV envelope. This prevents viral entry into the cells and disrupts viral replication.18
In a randomized, placebo-controlled clinical trial, docosanol in a 10% cream reduced the median time to healing of cold sores by 18 hours compared with a polyethylene glycol placebo cream. Among 737 healthy adult participants, treatment with the assigned product began during the prodromal or erythema stage of the HSL outbreak and continued 5 times daily until healing occurred. Healing was defined as the crust falling off spontaneously or the point when there was no evidence of an active lesion during twice-daily visits.19
The median time to healing in docosanol-treated patients was 4.08 days, which was significantly shorter than among the placebo-treated participants (4.80 days). Those in the active treatment group also had significantly shorter times from treatment initiation to cessation of pain and all other symptoms (median difference with docosanol, 13.4 h), complete healing of classic lesions (17.5 h), and cessation of the ulcer or soft crust stage of classic lesions (10.2 h). Approximately 20% of study participants reported at least 1 adverse effect during the study, but frequencies of specific reactions were not significantly different between the groups. Headache was the most common symptom, occurring in 5.9% of those in each treatment group. Application-site reaction occurred in 2.1% of docosanol-treated patients (versus 1.9% of those on placebo), 2.4% of those on active treatment had HSL lesions outside the treatment area (compared with 1.4% of placebo-treated participants, not a significant difference). All other adverse reactions were reported by fewer than 2% of those in each group.19
FDA-approved labeling for the marketed formulation of docosanol recommends initiating therapy in children aged 12 years or older or adults at the first sign of a tingle, bump, or itch that might indicate the start of an HSL outbreak and continuing treatment 5 times daily until lesions are healed. Patients should never share the pumps or tubes of docosanol with others because of the contagiousness of HSV (e.g., people who already get cold sores could be infected with other strains of HSV). Cosmetics can be applied over docosanol cream, but cotton-tipped, disposable applicators should be used to avoid contamination of the product with HSV. Because of potential hypersensitivity to docosanol, consumers should stop treatment if allergic symptoms occur, and those whose reactions become severe (e.g., hives, facial swelling, wheezing/difficulty breathing, shock, rash) should not use the product again. Patients whose HSL lesions are not healed within 10 days should seek medical care.20
Dietary supplements
Scant high-quality evidence is available for several alternative and complementary therapies that are labeled for prevention or treatment of cold sores. These include lysine products, tea tree oil, various vitamin and mineral combinations, and herbal products. Most such products are unstudied, and when trials are available, the study designs are weak or no significant improvements in patient symptoms have been reported.11,21,22
Pharmacist patient-care process
For consumers presenting in the pharmacy with a request for OTC therapy of cold sores, the pharmacist should confirm that the patient is immunocompetent and then ask about symptoms and look at any lesions that are visible. Patients with contraindications to OTC therapies should be referred for medical care; these include the presence of fever or swollen glands, rashes when docosanol has been used, lesions persisting after 10 to 14 days, and immunocompromised states, including those receiving chemotherapy or taking immunosuppressive drugs.
Pharmacists and other health or dental professionals who perform physical examination of patients’ mouths or potential HSL lesions should wear gloves at all times to avoid acquiring the virus or new HSV strains, spreading HSV to other patients, or auto-inoculation of other parts of their own bodies (as in the condition herpetic whitlow, which involves herpetic infection of the pulp at the end of the fingers). Consumers who participate in high-contact sports such as wrestling or soccer should also be aware of the possibility of transmission of the virus through such activities.3,23
If prodromal or visible symptoms have begun recently (within 1–2 days) in patients for whom self-care is appropriate, treatment with docosanol can be recommended. Based on consumer preference, symptomatic treatment with topical analgesics, cleansers, protectants, or antiseptics is reasonable. If lesions do not resolve in 10 days, consumers should seek medical evaluation of the lesions.20
In counseling patients seeking self-care products for cold sores, pharmacists should emphasize these points:
- With the antiviral agent docosanol, initiate therapy as quickly as possible when prodromal or visible symptoms occur. Patients who have several outbreaks each year should keep the product on hand at all times.
- Do not share topical HSL products since the virus is highly contagious and is likely present on the product packaging because of contact with lesions or the patient’s hands.
- If symptoms do not resolve in 10 days, systemic symptoms such as fever or rash appear, or immunocompetence is in doubt, the patient should seek medical evaluation and care.
- Advise patients who have numerous outbreaks each year that prescription antiviral agents are available for prophylactic or prompt systemic treatment of HSV.
COMMON PRESCRIPTION TREATMENTS FOR COLD SORES
Prescription topical, oral, or intravenous antiviral agents are sometimes used in the management of HSV infections. Knowing when patients with recurrent HSL should be referred for treatment with prescription agents is an important part of proper self-care advice.
Development of immunocompromised states necessitates more aggressive treatment of HSL, as outbreaks can become more frequent, persistent, and severe in these patients. Since prevention of outbreaks is an important therapeutic goal in immunosuppressed patients, long-term systemic therapy with oral or long-acting intravenous agents is needed.3 Given the self-care focus of this continuing pharmacy education program, management of immunocompromised patients is not covered in detail in this program.
Immunocompetent patients are also sometimes placed on prescription therapies, often with instructions to self-initiate therapy at the first sign or symptom of an outbreak. Since the virus cannot be eradicated, the goal of such oral therapies is to shorten or ameliorate the severity of lesions in an effort to lessen epithelial damage and spread of virus to close contacts.3
All currently available prescription antiviral agents are inhibitors of viral DNA synthesis. The first drug developed as an antiherpetic agent, acyclovir, is activated by a viral enzyme and acts by competing for a guanine moiety during DNA synthesis. Related medications are penciclovir, ganciclovir, foscarnet, and the prodrugs valacyclovir, famciclovir, and valganciclovir. Mutations that produce alterations in viral enzymes can lead to resistance to these antiviral agents.24
To be effective for HSL, topical agents must penetrate the epithelium and reach sites of viral replication in sensory nerve endings. Topical therapy is thus less effective than oral therapy, which provides systemic drug exposure, faster access to the sites of viral replication, greater bioavailability, less frequent dosing, and improved patient adherence.3,25
In a 2015 meta-analysis of 32 randomized controlled trials of 2640 people with recurrent cold sores, long-term use of oral antiviral drugs prevented cold sores, but the decrease in frequency was only 0.09 episodes per person per month. The efficacy of topical acyclovir cream applied to the lips was uncertain, and short-term use of oral or topical antiviral drugs did not prevent cold sores (Table 2).11,26-28 Adverse events did not differ between active and placebo therapies during short- or long-term use. The quality of evidence was low to moderate for most outcomes and very low for some outcomes.11
| Table 2. Interventions Tested for the Prevention of Cold Sores11,26-28 |
| Medication, length of therapy |
Clinical results |
| Acyclovir, oral, ≤1 month |
Inconsistent |
| Valacyclovir, oral, ≤1 month |
Uncertain |
| Famciclovir, oral, ≤1 month |
Uncertain |
| Acyclovir, oral, >1 month |
Reduced clinical recurrence |
| Valacyclovir, oral, >1 month versus episodic (when symptoms occur) |
Lower incidence of recurrence with long-term use than with episodic treatments |
| Acyclovir, topical, ≤1 month |
Little effect on recurrence |
| Penciclovir, topical, ≤1 month |
Reduced duration of lesions and pain by 0.5 d in 2 clinical trials of patients with recurrent cold sores |
| Foscarnet, topical, ≤1 month |
Little effect on prevention |
| Acyclovir, topical, >1 month |
Fewer research-diagnosed recurrences but no difference in participant-reported recurrences |
| 1,5-pentanediol, >1 month |
No preventive effect |
| 2-hydroxypropyl-beta-cyclo dextrin 20%, topical, >1 month |
Significantly more recurrences than with placebo |
| Sunscreen |
Prevented ultraviolet-induced recurrences in 1 trial; failed to reduce sunlight-induced recurrences in another |
Examples of topical regimens used in clinical practice for prevention or treatment of HSL outbreaks in immunocompetent adults include the following3:
- Acyclovir cream 5 times daily for 4 days (also approved for children aged 12 years or older)
- Penciclovir cream started at first symptom of cold sore and continued every 2 hours while awake for 4 days
Examples of oral regimens commonly used in clinical practice for chronic or intermittent suppressive therapy or treatment of HSL outbreaks include the following3,29:
- Acyclovir 200 mg to 400 mg 5 times daily for 5 days (treatment of outbreaks) or 400 mg 2 times daily (long-term suppression)
- Acyclovir buccal tablet 50 mg as a single dose applied within 1 hour of symptoms to the upper gum region and left in place for at least 6 hours
- Valacyclovir tablets 2 g every 12 hours for 1 day (FDA-approved dose); 500 mg twice daily for 5 days; 1 g once daily for 5 days
- Famciclovir 1000 mg twice daily for 1 day; initiate within 6 hours of symptom or lesion onset
When counseling patients who are receiving prescription therapies for cold sores, pharmacists should emphasize these points3,12:
- If the medication has been prescribed in anticipation of future HSL outbreaks, initiate therapy as quickly as possible when prodromal or visible symptoms begin.
- Remember to carry the medication on overnight trips, especially when the stresses of travel are a trigger for outbreaks.
- As soon as the medication supply is depleted, have the prescription refilled so it will be available for later episodes.
- Do not share topical products since the virus is highly contagious and is likely present on the product packaging because of contact with lesions or the patient’s hands.
- If symptoms do not resolve within 14 days after lesions appear or if systemic symptoms such as fever or rash appear or immunocompetence of the patient is in doubt, contact the prescriber or pharmacist.
THE ROLE OF THE PHARMACIST
The pharmacist is an important source of information for patients with oral lesions. As with self-care products in general, patients with HSL frequently rely on pharmacists for advice regarding treatment with OTC medications or seeking medical evaluation and prescription therapies.
As discussed previously, patients who have had HSV outbreaks often recognize prodromal symptoms of pruritus, tingling, and pain in the perioral area where fever blisters have appeared in the past. For maximal effectiveness, antiviral agents, either OTC or prescription, should be started as soon as such symptoms are noticed.
Pharmacists can inspect oral lesions when patients present with an outbreak already in progress; they should take care to wear gloves to avoid transfer of the virus to themselves or others. Cold sores are small, fluid-filled blisters occurring on the lips or nearby tissues; canker sores, occurring inside the mouth, are not caused by HSV and are managed differently than cold sores. Patients with cold sores who are immunocompromised and those with known risk factors for complications (atopic dermatitis, HIV infection, cancer) or taking immunosuppressive drugs should be referred for medical care.
Home care that can be recommended to appropriate patients is demonstrated in a video available from the AAD (https://www.aad.org/public/diseases/contagious-skin-diseases/herpes-simplex#tips). The tips are as follows30:
- Slow the outbreak: Burning, itching, or tingling may be the first sign that a cold sore is coming. When cold sores appear, apply an over-the-counter antiviral cream or ointment. Although this is not always effective, it may help slow the reproduction of the virus and relieve symptoms.
- Reduce pain: Consider taking ibuprofen or acetaminophen to help reduce pain.
- Avoid foods that contain acid. While you have a cold sore, avoid foods that contain acid, such as tomatoes and citrus fruits. These could irritate the skin and add to any pain.
- Cool the sores: Place a cool, wet towel on the cold sores for 5 to 10 minutes. Do this a few times daily to help reduce the redness and irritation.
Because of the increased risk of transmitting HSV to others during outbreaks, the AAD provides several points of advice and suggests taking special care when close contacts are at high risk (e.g., infants, immunosuppressed states). Until all cold sores have crusted over, patients should adhere to these precautions30:
- Do not kiss people, especially children.
- Avoid other intimate contact.
- Avoid close contact with anyone who has a weakened immune system, including newborn babies.
- Do not share lip balm, towels, razors, or other similar personal items.
- Do not share beverages or food.
- Try not to touch your cold sores.
- If you touch a cold sore or apply medicine to it, wash your hands immediately afterward.
- Wash your hands frequently throughout the day.
Pharmacists should counsel patients who are purchasing OTC products using the points provided in previous sections of this monograph. When referring patients for medical care for cold sores, reinforce the importance of having lesions evaluated to avoid potentially serious HSV outbreaks.
CONCLUSION
Pharmacists are one of the most accessible health professionals, providing a consultative role that is especially important for conditions such as cold sores. Pharmacists also offer a referral source for patients with HSV infections and outbreaks of cold sores. They are in a unique position to counsel patients regarding various treatments and can help optimize outcomes by recommending OTC products when those are appropriate and referring patients for prescription therapy as needed.
Currently available OTC products work through a variety of mechanisms of action that treat several aspects of HSL, including antiviral activity and symptoms of itching, pain, and tingling. For the majority of patients infected with this ubiquitous virus, OTC products play an important role in HSL management. Prescription products provide systemic therapy in those with frequent recurrences of HSL and risk factors that create a need to avoid HSL outbreaks.
RESOURCES
REFERENCES
- Herpesviruses. In: Carroll KC, Hobden JA, Miller S, et al, eds. Jawetz, Melnick, & Adelberg's Medical Microbiology. 27th ed. New York, NY: McGraw-Hill; 2016.
- Herpes simplex virus. World Health Organization. http://www.who.int/mediacentre/factsheets/fs400/en/#hsv1. Updated January 2017. Accessed August 1, 2017.
- Fatahzadeh M, Schwartz R. Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management. J Am Acad Dermatol. 2007;57(5):737-763.
- Cold sore. Mayo Clinic. http://www.mayoclinic.org/diseases-conditions/cold-sore/basics/definition/con-20021310. Updated May 15, 2015. Accessed August 1, 2017.
- Cunningham A, Griffiths P, Leone P, et al. Current management and recommendations for access to antiviral therapy of herpes labialis. J Clin Virol. 2012;53(1):6-11.
- What are cold sores? Academy of General Dentistry. http://www.knowyourteeth.com/infobites/abc/article/?abc=C&iid=298&aid=1149. Updated January 2012. Accessed August 16, 2017.
- Herpes simplex. American Academy of Dermatology. https://www.aad.org/public/diseases/contagious-skin-diseases/cold-sores. Accessed August 1, 2017.
- Patel R. Educational interventions and the prevention of herpes simplex virus transmission. Herpes. 2004;11(suppl 3):155A-160A.
- Mansell K. Bootsman N, Kuntz A, Taylor J. Evaluating pharmacist prescribing for minor ailments. Int J Pharm Pract. 2015;23(2):95-101.
- Worrall G. Herpes labialis. BMJ Clin Evid. 2009;pii:1704.
- Chi CC, Wang SH, Delamere FM, et al. Interventions for prevention of herpes simplex labialis (cold sores on the lips). Cochrane Database Syst Rev. 2015;(8):CD010095.
- Pray S. Preventing and treating cold sores. US Pharm. 2007;32(3):16-23.
- Hoheisel O. The effects of Herstat (3% propolis ointment ACF) application in cold sores: a double-blind placebo-controlled clinical trial. J Clin Res. 2001;4:65-75.
- Hasan T, Rantanen T, Alanko K, et al. Patch test reactions to cosmetic allergens in 1995-1997 and 2000-2002 in Finland—a multicentre study. Contact Derm. 2005;53(1):40-45.
- Kaminester LH, Pariser RJ, Pariser DM, et al. A double-blind, placebo-controlled study of topical tetracaine in the treatment of herpes labialis. J Am Acad Dermatol. 1999;41(6):996-1001.
- Herpes simplex virus. University of Maryland Medical Center. http://www.umm.edu/health/medical/altmed/condition/herpes-simplex-virus. Updated September 29, 2015. Accessed August 16, 2017.
- Dermatologists share tips for treating cold sores. American Academy of Dermatology. https://www.aad.org/media/news-releases/dermatologists-share-tips-for-treating-cold-sores. Updated April 15, 2015. Accessed August 1, 2017.
- Safrin S. Antiviral agents. In: Katzung BG, Trevor AJ, eds. Basic & Clinical Pharmacology. 13th ed. New York, NY: McGraw-Hill; 2015.
- Sacks SL, Thisted RA, Jones TM, et al; Docosanol 10% Cream Study Group. Clinical efficacy of topical docosanol 10% cream for herpes simplex labialis: a multicenter, randomized, placebo-controlled trial. J Am Acad Dermatol. 2001;45(2):222-230.
- Abreva [product labeling]. Warren, NJ: GSK Consumer Healthcare;2008.
- Tomblin FA Jr, Lucas KH. Lysine for management of herpes labialis. Am J Health Syst Pharm. 2001;58(4):298-300,304.
- Singh BB, Udani J, Vinjamury SP, et al. Safety and effectiveness of an L-lysine, zinc, and herbal-based product on the treatment of facial and circumoral herpes. Altern Med Rev. 2005;10(2):123-127.
- Wheeler CE Jr. The herpes simplex problem. J Am Acad Dermatol. 1988;18(1 pt 2):163-168.
- Antiviral agents (nonretroviral). In: Hilal-Dandan R, Brunton LL, eds. Goodman and Gilman's Manual of Pharmacology and Therapeutics. 2nd ed. New York, NY: McGraw-Hill; 2014.
- Esmann J. The many challenges of facial herpes simplex virus infection. J Antimicrob Chemother. 2001;47 Suppl T1:17-27.
- Boon R, Goodman JJ, Martinez J, et al. Penciclovir cream for the treatment of sunlight-induced herpes simplex labialis: a randomized, double-blind, placebo-controlled trial. Penciclovir Cream Herpes Labialis Study Group. Clin Ther. 2000;22(1):76-90.
- Spruance SL, Rea TL, Thoming C, et al. Penciclovir cream for the treatment of herpes simplex labialis. A randomized, multicenter, double-blind, placebo-controlled trial. Topical Penciclovir Collaborative Study Group. JAMA. 1997;277(17):1374-1379.
- Denavir [package insert]. Wehr, Germany: Prestium Pharma, Inc.;2013.
- Cernik C, Gallina K, Brodell RT. The treatment of herpes simplex infections: an evidence-based review. Arch Intern Med. 2008;168(11):1137-1144.
- Herpes simplex: tips for managing. American Academy of Dermatology. https://www.aad.org/public/diseases/contagious-skin-diseases/herpes-simplex#tips. Accessed August 24, 2017.
Back to Top