
Expired activity
Please go to the PowerPak
homepage and select a course.
How Specialty Pharmacists Can Enhance Patient-Driven Care in Multiple Sclerosis-Article
ROLE OF SPECIALTY AND MANAGED CARE PHARMACISTS IN MS
Specialty and managed care pharmacists are increasingly involved in the management of
multiple sclerosis (MS), as the number of approved biologic therapies expands, more
intravenous (IV) and specialty formulations are approved, and the cost of treatment continues
to rise. Specialty pharmacists play important decision-making roles in managing high-cost,
serious diseases such as MS and may be involved in recommending a course of therapy, setting
up monitoring parameters, and providing patient counseling to address barriers in adherence to
and persistence with medications used in this disease.1,2
To participate in formulary decision-making and authorization for initial therapy and
therapeutic switches, pharmacists in managed care settings need thorough knowledge of the
approved and emerging MS disease-modifying therapies (DMTs), their contraindications and
safety warnings, and other relevant aspects of care.3 As more risk-intensive therapies are
introduced, pharmacists are often called to evaluate therapeutic risks and benefits. Finally,
pharmacists in many practice settings need strategies for patient counseling in this highly
complex and rapidly changing therapeutic area.
Keeping up with the latest changes in MS therapeutics can be logistically difficult even for
clinicians who have extensive experience with MS. On a national level, there is a demand for
management of chronic conditions such as MS to be more patient-centered.4,5 Engaging the
patient in shared decision-making about the healthcare process has been shown to have an
impact on adherence to treatment, and to prevent outcomes such as avoidable
hospitalizations, emergency department visits, and unnecessary complications.5,6
MS OVERVIEW: MS DISEASE PRESENTATION AND PHENOTYPES
MS is a chronic, disabling degenerative disease characterized by central nervous system (CNS)
inflammation, presumably linked to autoimmunity.7 Over time, breakdown of the myelin
sheaths surrounding nerve fibers in the brain and spinal cord leads to scarring, axonal loss, and brain atrophy. Symptoms include muscle weakness and/or spasticity, visual deficits, bladder
and bowel dysfunction, fatigue, and cognitive impairment.7
Relapse occurs when a loss of integrity in the blood-brain barrier allows infiltration of
inflammatory cells and cytokines that target myelin, axons, neurons, and oligodendrocytes. The
resulting inflammatory cascade activates microglial cells, resulting in the loss of neurons and
axons and the enhanced or new white matter lesions observed on gadolinium-enhanced MRI.8,9 Disability results when recovery from relapse is incomplete. Relapse is a clear sign of
inadequate disease control, with many studies finding frequent relapses in the first 2 to 5 years
correlated with disease worsening.10,11
Forms of MS
MS presents in different forms, which have been redefined somewhat over the years. The 3
most commonly recognized forms of MS are diagrammed in Figure 1.12 Most cases of MS
(about 80%) present as a relapsing-remitting course (RRMS) characterized by acute relapses or
exacerbations, followed by periods of remission which may involve a return to baseline
symptomatically, especially early in the disease course. Over time, incomplete recovery from
relapses causes progression of disability and may lead to a secondary progressive phase (SPMS).
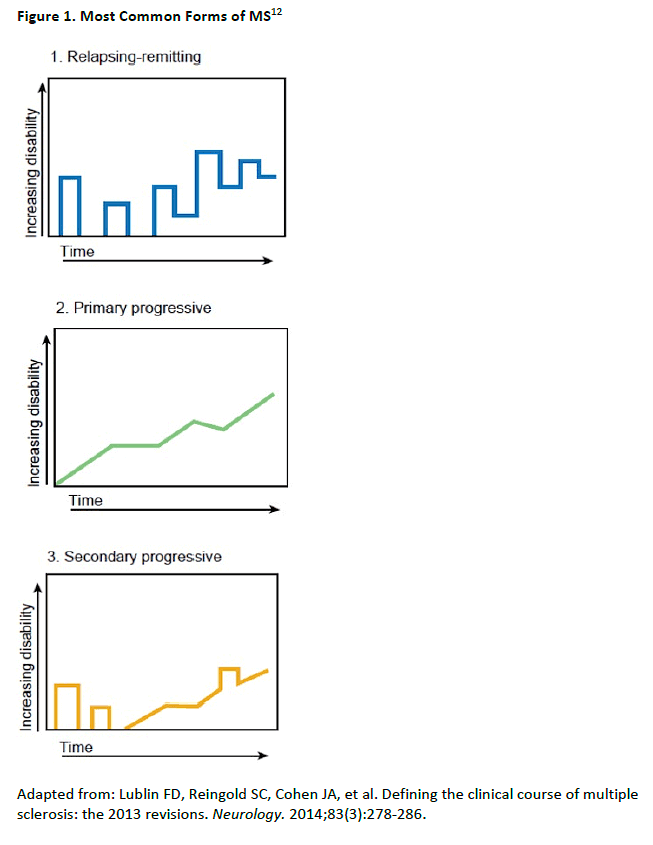
About 10% to 15% of patients with present initially with a primary progressive course (PPMS) in
which there is progression of disease from onset.12 Some of these patients may also have
superimposed relapses indicating active inflammation (about 6% to 10% of those with PPMS),
but their condition does not return to baseline, instead showing a steady trajectory of
disability.13
Patients with SPMS start out with RRMS but as the disease progresses it appears to shift from a
condition driven mainly by inflammation, to one that is primarily degenerative. DMT, especially
early in the RRMS phase, is known to delay onset of SPMS by several years in a high percentage
of patients.14
MS disease presentation is highly heterogeneous, and within these classifications other
phenotypes have been identified to better characterize the pattern of inflammatory activity
which may correlate to a patient's response to therapy. The most recent phenotypic
classification for MS is outlined in Table 1.12
| Table 1. MS Phenotypes, 2013 Classification12 |
| Active disease |
- Clinical: relapses, acute or subacute episodes of new or increasing neurologic dysfunction followed by full or partial recovery, in the absence of fever or infection and/or
- Imaging (MRI): occurrence of contrast-enhancing T1 hyperintense or new or unequivocally enlarging T2 hyperintense lesions
|
| Progressive disease |
- Clinical: steadily increasing objectively documented neurologic dysfunction/disability without unequivocal recovery (fluctuations and phases of stability may occur)
- Imaging (MRI): imaging measures of progression are not established or standardized and not (yet) useful as phenotype descriptors for individual patients. Under consideration are increasing number and volume of T1-hypointense lesions, brain volume loss, and changes in magnetic transfer imaging and diffusion tensor imaging
|
| Worsening disease |
- Documented increase in neurologic dysfunction/ disability as a result of relapses or progressive disease, reserving the term “disease progression” for those solely in a progressive phase of the illness, who are gradually worsening
|
| Confirmed progression or worsening |
- Increase of neurologic dysfunction confirmed throughout a defined time interval (for example, 3, 6, or 12 months)
|
| Source: Lublin FD, Reingold SC, Cohen JA, et al. Defining the clinical course of multiple sclerosis: the 2013 revisions. Neurology. 2014;83(3):278-286.35 Reprinted through Open Access/Creative Commons License 3.0. |
UPDATE ON MS DISEASE-MODIFYING THERAPIES
It is an ongoing challenge to keep up with rapid changes in the field of MS as newer therapies
are introduced, many with novel mechanisms of action and a wider range of risks not previously
associated with treating this disease. As of January 2018, there were a total of 14 DMTs
approved by the U.S. Food & Drug Administration (FDA) for treatment of MS.15 (This list does not
include mitoxantrone and cyclophosphamide, which are utilized less frequently in the U.S. due
to toxicity risk).16 Table 2 begins with injectable therapies, which include the original "platform"
therapies, interferon beta-1a (IFNB-1a), interferon beta-1b (IFNB-1b), and glatiramer acetate
(GA).15 Recently, generic formulation for GA were introduced for both the 20 mg and 40 mg
doses.17 The newest injectable agent is daclizumab, approved in 2016 for treatment-refractory
MS and administered on a once-monthly dosage schedule.18
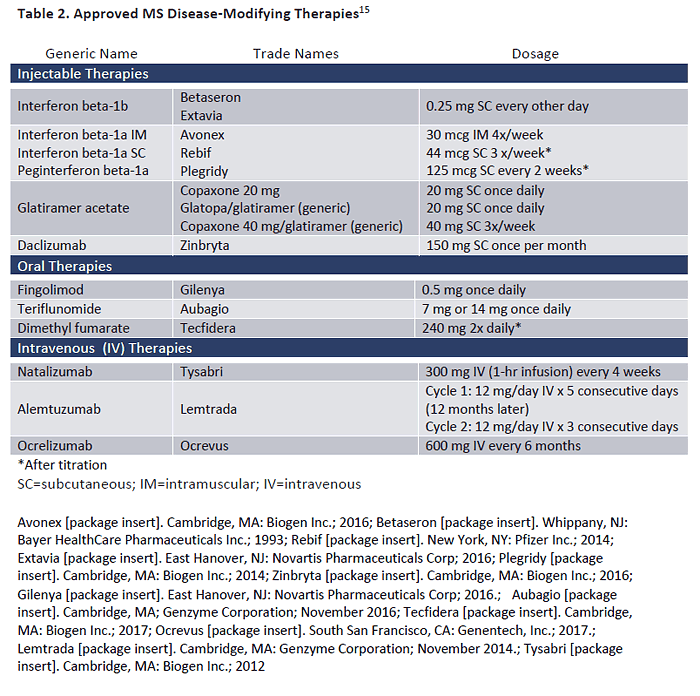
Oral DMTs for MS first became available in 2010. The currently available oral agents are also
shown in Table 2.15 Oral agents offer an administration advantage for patients but, as
pharmacists know, this does not always translate to advantages in tolerability or adherence.19,20 Some of the oral DMTs (e.g., dimethyl fumarate) recommend dosage titration to minimize
adverse effects (AEs). In addition, some (such as fingolimod) involve a number of pre-dose
screening and monitoring steps. For patients receiving teriflunomide, it is essential to ensure
effective birth control for female patients due to its Category X pregnancy rating.21
Approved IV therapies for MS are also shown in Table 2.15 Natalizumab (administered IV every 4
weeks) was approved in 2004, removed from the market due to emergence of progressive
multifocal leukoencephalopathy (PML). It was later re-introduced in 2006 under a risk
evaluation and mitigation strategy (REMS) program.22 Alemtuzumab is given in 2 cycles: the first
cycle over 5 consecutive days, and then a year later for 3 consecutive days. Longer-term
research has suggested that some patients do not require subsequent doses, but it remains in
question whether patients should be switched to a maintenance therapy with another DMT
following alemtuzumab treatment.23 Ocrelizumab, a humanized form of the CD20-targeted
monoclonal antibody used in rituximab, was approved for MS in March 2017.24
Pipeline: Experimental and Emerging Therapies
As new agents are introduced for the management of MS, formulary and benefit decisions need
to be re-evaluated. The current MS pipeline has a wide variety of newer agents under
investigation, some of which utilize novel mechanisms of action.25 As of early 2018, the oral
agent cladribine is under FDA review for MS. This agent is approved in the U.S. for hairy cell
leukemia and was recently approved in Europe and Canada as a second-line therapy for MS.26,27 Also in the pipeline are newer, selective S1P1 receptor modulators (e.g., siponimod,
ponesimod).25,28 Ofatumumab is a monoclonal antibody targeting lymphocytes that express
CD20. This agent is approved for chronic lymphocytic leukemia (CLL) in the U.S. and is in clinical
trials for MS.29
THERAPEUTIC GOALS AND SHARED DECISION-MAKING IN MS
The goals of therapy in MS involve a balance between efficacy in controlling the disease, and
optimizing safety and tolerability (Figure 2). Efficacy outcomes in MS trials include:30-32
- Annualized relapse rate (ARR)
- MRI lesions (number, size, and location, change from baseline)
- New/enlarging T2 or gadolinium-enhancing lesions
- Brain atrophy (algorithms measuring whole brain volume, change from baseline)
- Disability progression, often measured via Expanded Disability Status Scale (EDSS) or MS Functional Composite (MSFC)
- “NEDA" (no evidence of disease activity)
— No sustained disability in 3 months
— No relapses
— No MRI activity
Although we often think of treating MS in terms of reducing relapses (ARR), it is important to
consider other efficacy goals such as reducing MRI lesion burden and progression of disability.
Disability reduction can be difficult to demonstrate in short-term trials and often does not
correlate with other efficacy outcomes.33 In clinical trials disability, is usually measured via
EDSS, although this tool is used less often in clinical practice. Brain atrophy is another measure
used in clinical trials to evaluate DMT treatment effect, but not often used in clinical practice. A
newer goal that is akin to remission is NEDA, which stands for “no evidence of disease activity”
using the criteria listed above.34-36 Safety outcomes include frequency of AEs, including
tolerability and serious AEs such as infection, malignancy, and liver toxicity.22
When balancing safety and efficacy in MS, a key issue emphasized by major MS organizations is
the need for shared decision-making with the patient.37 Because the presentation and course of
MS vary so widely among individuals, it is not possible to predict which DMT will work best for a
given patient, supporting the need for individualization of therapy. Some of the more recently
introduced therapies offer superior efficacy outcomes (e.g., low ARR) but also carry a higher
risk of serious AEs.22 Patients whose disease presentation is more aggressive, with extensive
inflammatory activity, warrant a more aggressive treatment approach.38,39 In addition, it is
important to take into consideration the patient's tolerance for risk and willingness to accept
the potential for a serious AE.40
For many patients, the mode of administration (e.g., IV, subcutaneous, or oral) is one of the
overriding factors in their decision to start a particular MS therapy. Are they open to giving
themselves injections? If so, are they physically capable of doing so, or have a care partner who
can help them? Are they prepared to keep infusion center appointments and maintain the
steps in the REMS? Another issue to consider is the need to keep up with required monitoring
steps, which in some cases may mean pre-dose monitoring and/or monthly blood tests.41
What Are the Current Guideline Recommendations?
Official guideline statement for disease-modifying therapy in MS have been lacking for many
years. This year, two major guideline statements are anticipated, a joint statement from two
European MS organizations—the European Committee for Research and Treatment of Multiple
Sclerosis (ECTRIMS) and European Academy of Neurology (EAN)—and a statement from the
American Academy of Neurology (AAN) both expected to be released in early 2018.42,43
Comparing MS Therapies
Managed care and specialty pharmacists frequently participate in clinical evaluations and
formulary decisions related to the rapidly evolving field of MS management.44-48 This may
require the formulary team to evaluate and compare MS DMTs, yet these medications are
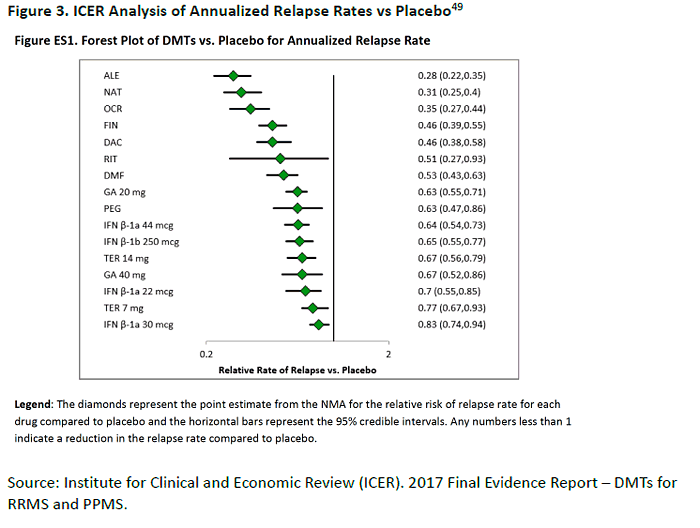
difficult to "rank" or compare head-to-head. A 2017 Institute for Clinical and Economic Review
(ICER) report summarized multiple network meta-analyses to compare the available therapies
on various criteria, including ARR, price, and quality-adjusted life years (QALY).49 As shown in
the Forest plot in Figure 3, alemtuzumab, natalizumab, and ocrelizumab had the greatest
reduction in ARR (approximately 70% reduction, compared to placebo).49 Fingolimod,
daclizumab, rituximab, and dimethyl fumarate were next, with 47% to 54% reduction rates.
(The meta-analysis included trials of rituximab, used off-label.) Within these groupings,
however, 95% confidence intervals overlapped to some degree.49 The interferons, glatiramer
acetate 20 mg, and teriflunomide showed 17% to 37% ARR reductions. One caveat to keep in
mind is that most of the trials in the analysis were not conducted head-to-head, with key
differences in entry criteria, trial designs, and baseline study population. It has been argued that the older studies (interferons and glatiramer acetate) enrolled patients who had more
severe disease, due to older diagnostic criteria.50 In addition, ARR is only one of the efficacy
criteria on which MS drugs are evaluated.
Starting Therapy: Effects of Early Versus Delayed
Multiple MS studies support the benefits of starting therapy early in the course of the disease,
which translates to improved outcomes over time. Long-term follow-up studies of several
pivotal trials show that patients started on the active treatment do better than those started on
placebo, with these benefits sustained over many years. These benefits include lower ARR,
longer time periods before the onset of SPMS, retention of employment, and improved quality
of life.33,51-53 Long-term data also support a survival benefit for patients treated early compared
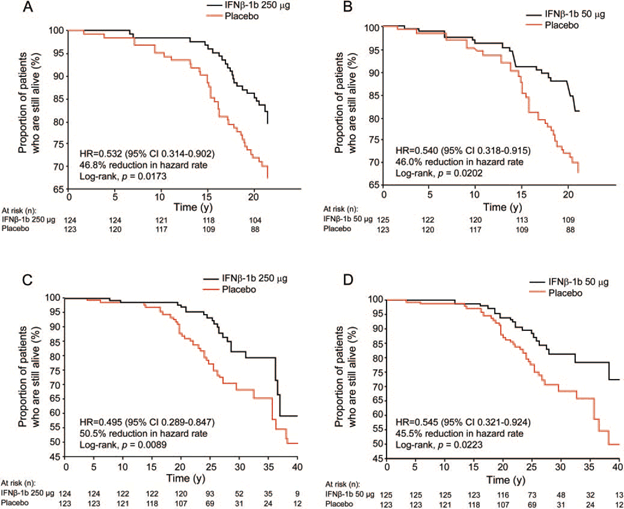
with those whose treatment was delayed. A 21-year follow-up study from the pivotal IFNB-1b
trials (with 98% ascertainment rate) showed that patients started on the active treatment had a 46.8% decrease in all-cause mortality compared with those who were started on placebo (Figure 4).54 In this study patients treated early had improved survival regardless of the
presence of other indicators of poor prognosis.13,33,54,55
Figure 4. Survival from pivotal randomized controlled trial randomization over 21 years is shown for interferon β-1b (IFNB-1b) 250g vs placebo (A) and IFNB-1b 50g vs placebo (B). Time from onset of clinical symptoms to death is shown for IFNB-1b 250 g vs placebo (C) and IFNB-1b 50 g vs placebo (D). Hazard ratios (HRs) and 95% confidence intervals (CIs) are estimated using Cox proportional hazard models without stratification.
|
| Reprinted with permission from: Goodin DS, et al. Neurology. 2012;78:1315-1322. |
In contrast, many patients who delay treatment during the critical "window of time" early in the
disease never really quite catch up, and have a significantly shorter time to before reaching
disability milestones such as need for a cane or wheelchair and onset of SPMS.53
SAFETY MONITORING OF MS AGENTS
An important part of the pharmacist’s role is detecting potential contraindications or adverse
event risks that could compromise medication safety.56,57 Specialty and managed care
pharmacists need thorough knowledge of the contraindications, safety warnings, and REMS programs for the MS DMTs. Pharmacists also need strategies for patient counseling in this
highly complex and rapidly changing therapeutic area.
The need to balance efficacy in MS treatment with safety and risk avoidance has grown more
complex as the number of immunomodulators for MS has increased.58,59 Risks of serious
infections, autoimmune liver and thyroid dysfunction, cardiovascular risks, and teratogenicity
must be balanced with the significant health risks associated with progression of the MS
disease process.29,56,60,61 However, one must not overlook the significant health risks associated
with progression of the MS disease process itself.56 Current REMS programs for the MS
therapies include:
- TOUCH Prescribing Program for natalizumab (Tysabri)
- LEMTRADA REMS Program (alemtuzumab)
- ZINBRYTA REMS Program (daclizumab)
A REMS program previously in place for fingolimod (Gilenya) was discontinued by the FDA as of
December 2016.62
Cancer and Malignancies
The question of a link between MS therapies and malignancy has been debated, but in most
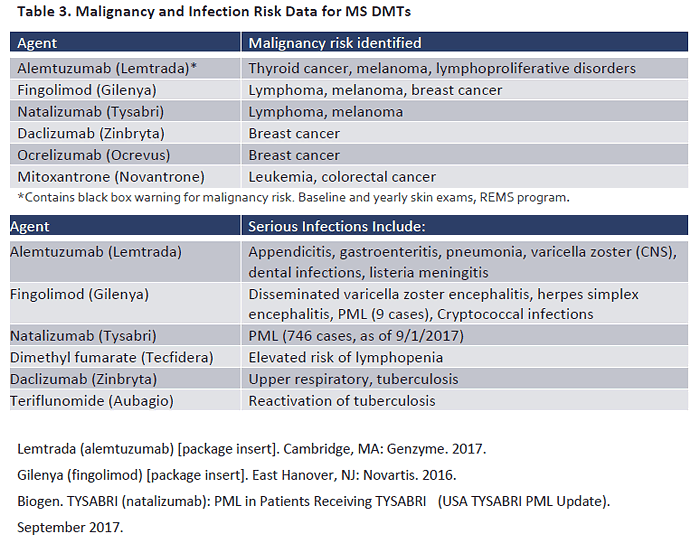
controlled studies malignancy rates are low and causation has not been determined.63,64 Table 3 summarizes current data on malignancy risk reported in the labeling and literature.65,66 The
labeling for alemtuzumab contains a black box warning that calls for baseline and yearly skin
exams to detect malignancies such as melanoma.67
Infection Risks with MS DMTs
The MS drugs modify or suppress the immune system by modulating or depleting T cells and/or
B cells, which leaves patients at risk for infection.68 Table 3 reviews infection types reported in
controlled trials of selected MS therapies. The most serious infection risk is for PML, associated primarily with natalizumab. PML is a serious brain infection that is highly fatal and—for patients
who survive it—highly debilitating. As of Fall 2017 there were 746 confirmed PML cases among
patients using this agent.69 Although there are a handful of cases with the other drugs, they do
not reach this magnitude and some might be attributed to prior natalizumab use.70 However,
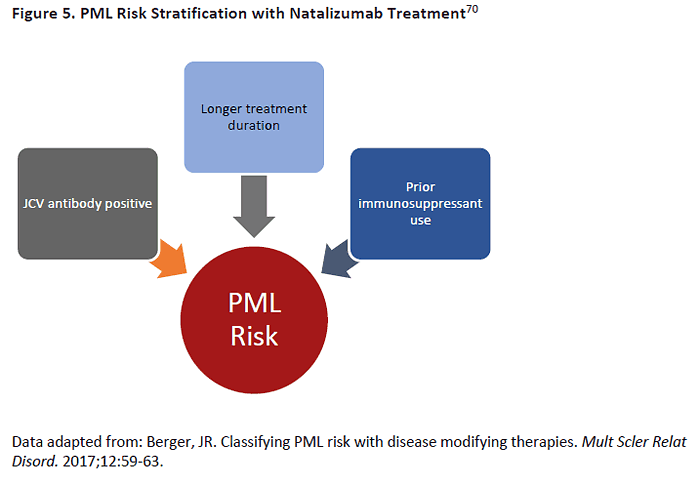
we can limit PML risk significantly, mainly by limiting natalizumab use to patients who do not
have antibodies for John Cunningham virus (JCV) (Figure 5). The risk of PML per 1,000 patients
treated is minimal for people who are JCV negative, regardless of how long they have received
natalizumab. Those who are JCV positive but have not used immunosuppressant drugs have a
low risk (<1 per 1,000 treated patients) if treated with natalizumab for less than 2 years, but
this risk starts to climb after 4 years (estimated 3 cases per 1,000 treated) and 6 years (6 cases
per 1,000).69 Among patients who are both JCV positive and have prior immunosuppressant
use, the risk is low if treated under 2 years (1 per 1,000) but jumps significantly to 12 or 13
cases per 1,000 for those treated for 4 years or longer. For these reasons, many neurologists
will switch patients to another agent if they seroconvert to JCV positive status and have been
on treatment for more than 2 years.69
Secondary autoimmunity is another AE category that may be observed in MS, particularly
associated with the use of alemtuzumab. While this treatment is highly efficacious, thyroid AEs
affect a high percentage of patients treated with alemtuzumab. In the CARE-MS I trial of
treatment-naive patients, 18.1% of alemtuzumab-treated patients developed thyroid-related
AEs, compared with 6.4% of those receiving interferon. In the CARE-MS II trial (patients who
had disease activity while on another DMT) thyroid complications occurred in 15.9% versus 5%
for the interferon.71,72
Safety Monitoring Steps
Pharmacists in all practice areas provide a crucial role in the prevention of medication errors
and avoidable adverse events.73,74 Pharmacists may alert patients to possible medication risks,
and clarify with the provider about whether a dosage is correct or a comorbidity should be of concern. For patients with MS, the pharmacist may have a role in ensuring that the required or
recommended monitoring steps are met for these therapies before a patient receives an initial
prescription or authorization for a refill. In addition, pharmacists are frequently in a position to
remind and educate patients and other health care providers about the need for monitoring.
Recommended monitoring steps (adapted from product labeling) are summarized in Table 4.
| Table 4. Monitoring Steps for MS DMTs |
| Injectable agents |
Key Monitoring Steps |
Interferon b-1a (Avonex, Rebif)
Interferon b-1b (Betaseron, Extavia)
Peginterferon beta-1a (Plegridy) |
CBC, LFT
Skin examination
Thyroid hormone levels |
| Glatiramer acetate (Copaxone) |
Skin examination |
| Daclizumab (Zinbryta) |
Predose: Serum transaminases and total bilirubin; screen for TB, Hep B and C, active infections
Post-dose: Monitor liver enzymes periodically |
| Oral agents |
Key Monitoring Steps |
|
| Fingolimod (Gilenya) |
ECG monitoring for bradycardia on initial dosing
Ophthalmic examinations – macular edema
CBC, LFT |
|
| Teriflunomide (Aubagio) |
Pregnancy test/counsel about risk of pregnancy
Monthly LFT for first 6 months
Tuberculosis skin testing |
|
| Dimethyl fumarate (Tecfidera) |
CBC with lymphocyte count prior to initiating therapy
CBC with differential every 6 months |
|
| Infused agents |
Key Monitoring Steps |
| Ocrelizumab (Ocrevus) |
• Monitor for infusion reactions
• Delay treatment in patients with active infections
• Avoid vaccination with live-attenuated vaccines during and soon after treatment |
Alemtuzumab (Lemtrada)
(Restricted distribution program) |
• Pre-dose: re-medicate with corticosteroids for first 3 days of course
• Use herpetic prophylaxis on first day of dosing, continue for ≥ 2 months after dosing
• Obtain thyroid function tests prior to treatment
• During infusion: Monitor for infusion reactions including serious reactions
• Post-dose: Increased risk of malignancies. Perform baseline and yearly skin exams
• Obtain thyroid function tests every 3 months until 48 months after last dose
• Delay treatment in patients with active infections |
Natalizumab (Tysabri)
(Restricted distribution program) |
• Monitor for risk of PML: obtain JC virus status and other PML risk factors
• Monitor for hepatotoxicity
• Monitor for symptoms of hypersensitivity
• Monitor for infections
• TOUCH Prescribing Program: evaluate 3 and 6 months after first infusion and every 6 months thereafter, and for ≥6 months after discontinuing.
• Determine every 6 months whether patients should continue on treatment. |
| Avonex [package insert]. Cambridge, MA: Biogen Inc.; 2016; Betaseron [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc.; 1993; Rebif [package insert]. New York, NY: Pfizer Inc.; 2014; Extavia [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2016; Plegridy [package insert]. Cambridge, MA: Biogen Inc.; 2014; Zinbryta [package insert]. Cambridge, MA: Biogen Inc.; 2016; Gilenya [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2016.; Aubagio [package insert]. Cambridge, MA; Genzyme Corporation; November 2016; Tecfidera [package insert]. Cambridge, MA: Biogen Inc.; 2017; Ocrevus [package insert]. South San Francisco, CA: Genentech, Inc.; 2017.; Lemtrada [package insert]. Cambridge, MA: Genzyme Corporation; November 2014.; Tysabri [package insert]. Cambridge, MA: Biogen Inc.; 2012 |
INDICATIONS FOR SWITCHING THERAPIES
Switching therapies is another complex and controversial aspect of MS therapy. In general,
switching therapies is recommended if a patient has a suboptimal response to a DMT including
relapse activity, increase in symptoms, or increased MRI activity.60 Switches are also authorized
for safety risks (including laboratory abnormalities) and tolerability problems, which could
include flu-like AEs associated with the interferons or injection-site reactions.75 Another
possible reason for switching is antibody development, which affects mainly natalizumab but
also the interferons.75,76 Table 5 groups some of the recognized medical indications for
switching.75
Table 5. Medical Indications for Switching Therapies75
| Indication |
Category |
Examples |
| Intolerability |
Adverse reactions |
Injection site reaction, infusion reaction, infections |
| Persistent symptoms |
Flu-like symptoms, headache, nausea |
| Significant and persistent laboratory abnormality |
Increased liver enzymes, low WBC |
| Detection of antibodies |
JCV Ab+ |
Natalizumab |
| Persistent neutralizing antibodies |
Natalizumab, interferons |
| Unacceptable disease activity |
Clinical activity |
Relapses, disability, cognitive status, transition to progressive disease |
| Neuroimaging activity |
Brain/spinal cord MRI abnormalities |
| New therapy options |
Safer, more efficacious therapies become available |
WBC=white blood cells; JCV=John Cunningham virus
Data adapted from: Coyle PK. CNS Drugs. 2013;27(4):239-247.
Data from a variety of patient surveys describe the reasons given by patients for requesting a
therapeutic switch. For example, a 2010 study by Reynolds et al of 5,722 patients with MS in a
managed-care setting revealed that between 17% and 41% of patients ultimately discontinue
DMT, most within the first 2 years. This study showed high rates of switching, with 75% of
patients switching 1 time, 11% switching twice, and 14% switching 3 or more times.77
In a survey conducted by the AAN among patients with MS who switched therapies, the main
reasons given were:78
- Desire to try a newer drug or different mode of administration
- Injection fatigue, injection-site reactions
- Feeling that therapy is not working for them (efficacy/disease severity)
- Nonadherence (may not be reported or obvious)
- Side effects/tolerability
- Concerns about risks of therapy
- Life changes (e.g., pregnancy)
- Comorbidities
Data from the "MS in America" survey from 2014 involving over 5,000 patients with MS
confirmed that “injection fatigue” or “needle fatigue” was at the top of the list of reasons for
desiring a switch.79
Some managed care companies mandate that patients be on a therapy for a set period of time
before switching, or present evidence that the therapy is associated with breakthrough disease
or tolerability problems. However, it should be noted that because MS is a slowly progressing
disease, efficacy response to a new treatment may take several months or even up to a year to
evaluate. However, if a patient has new MRI lesions or an MS relapse while on therapy, this is
often a prompt to re-evaluate the patient for a possible therapeutic switch, usually to a
different class of agent. Nonadherence should be considered as a possible cause of suboptimal
response.80 With a greater variety of treatment options available, we can expect an increase in
the frequency of switching.
COSTS OF MS THERAPIES
Healthcare providers and patients cannot overlook the economic realities involved in MS
management. Regardless of which agent is used, medications for MS are high-cost agents that
make up a significant portion of the specialty pharmacy budget and payers’ outlays for chronic-care medications.30,81-85 A study published in 2015 calculated that DMTs costing between
$8,000 and $11,000 per year approximately 20 years ago now cost over $70,000 per year.86 Table 6 cites Year 1 acquisition costs for DMTs as reported in the ICER report from 2017.49 As
the cost of MS DMTs has steadily increased, managed care plans and other payers have introduced restrictions on how and when these agents are prescribed. It is essential that
pharmacists in managed care and specialty pharmacy settings are equipped for their role in
initiating DMT, maintaining therapy, and receiving the appropriate support and reimbursement.
Table 6. Year 1 Acquisition Costs of MS DMTs49
Injectable
| Interferon B-1a (Avonex) |
$81,965 |
| Interferon B-1a (Rebif) |
$86,416 |
| Interferon B-1b (Betaseron) |
$86,659 |
| Interferon B-1b (Extavia) |
$72,359 |
| Peginterferon B-1a (Plegridy) |
$81,956 |
| Glatiramer acetate (Copaxone) 40 mg |
$86,554 |
| Glatiramer acetate (Copaxone) 20 mg |
$76,024 |
| Glatiramer acetate (Glatopa) 20 mg |
$63,193 |
| Daclizumab (Zinbryta) |
$82,000 |
Oral
| Fingolimod (Gilenya) |
$82,043 |
| Teriflunomide (Aubagio) |
$76,612 |
| Dimethyl fumarate (Tecfidera) |
$82,977 |
IV Infusion
| Natalizumab (Tysabri) |
$78,214 |
| Alemtuzumab (Lemtrada) |
$103,749 |
| Ocrelizumab (Ocrevus) |
$65,000* |
Source: Institute for Clinical and Economic Review (ICER). 2017 Final Evidence Report – DMTs for RRMS and PPMS.
*Price from Red Book online accessed 9-2017 |
Pharmacoeconomic Issues in MS
Patients with MS want the option of working with their health care provider to choose the DMT
that is best for them, but may feel that their choice of therapy is driven by insurance coverage
and the willingness of their provider to appeal denials of coverage.49 Survey results included in
the 2017 ICER report show that many patients with MS believe they are limited in their
treatment choices due to financial and coverage barriers. In a survey of 16,000 patients with
MS, 69% said health plan restrictions affected their therapeutic choices, and 66% said that out
of pocket costs impacted their treatment decisions.87
Patients and even many providers may be unaware of the true costs of MS therapies, due to
continual price increases, copay assistance programs, and different pricing structures that may
be negotiated between the payer and the manufacturer. Many managed care companies often
look at costs in terms of per member/per month (PMPM) calculations. According to the most
recent published trend report from Express Scripts, the PMPM for MS was $5.00 for pharmacy
benefits in 2016, which is in addition to what is spent on pharmacologic therapies such as
natalizumab. This means that every member (adult and child) in the health plan must
contribute $5 per month to cover the pharmacy benefit costs of DMTs for patients with MS in
the plan.88
Most specialty pharmacies have robust programs in place to help patients with their out-ofpocket (OOP) costs. These programs work in conjunction with those offered by pharmaceutical
companies and other organizations. In fact, most MS drug manufacturers offer patient
assistance programs that provide full or partial coverage or co-pay assistance. Most require
financial and coverage documentation and many require re-enrollment after a certain time period. For example, in Accredo's specialty pharmacy program 98% of specialty drugs have an
available copay assistance program ($364 million in total assistance) and the specialty
pharmacy provides services to connects payers and patients for copay assistance.89
Economic analyses in MS are controversial because of the mismatch between cost analysis
measures (such as cost per relapse avoided) and the difficulty of quantifying the value of
providing DMT. Many analyses use QALY thresholds of up to $150,000 to consider whether an
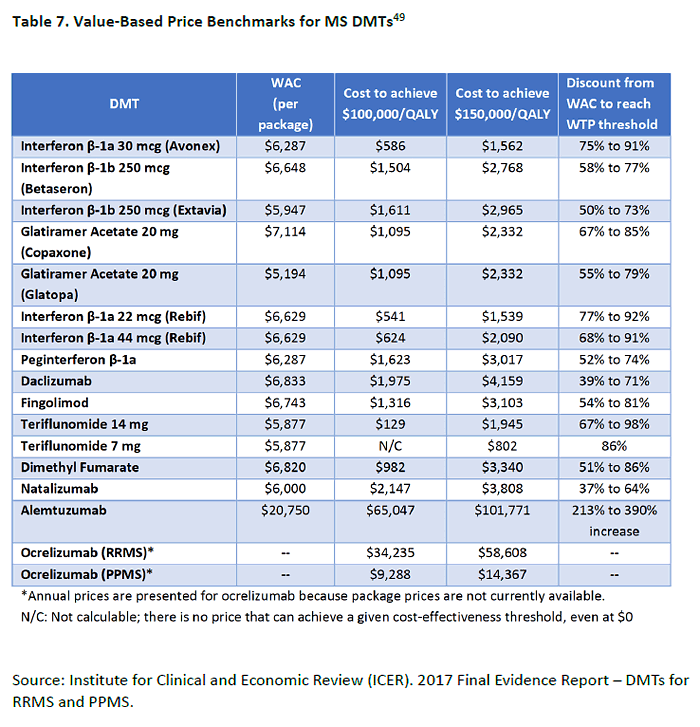
intervention can be regarded as cost effective. For example, an economic analysis in the ICER
report shows that the majority of DMTs would need to have a 37% to 98% discount off their
wholesale acquisition cost (WAC) to meet thresholds for cost effectiveness or society's
"willingness to pay" (Table 7). The higher dose of teriflunomide would need a WAC around
$2,000 to make the threshold, while ocrelizumab could be priced around $59,000 based on
higher efficacy ratings.49
PATIENT EDUCATION AND SUPPORT
There is a demand on a national level that management of chronic conditions, including MS,
become more patient-centered.90,91 Engaging the patient in the healthcare process has been
shown to have an impact on adherence to treatment, and to prevent outcomes such as
avoidable hospitalizations, emergency department visits, and unnecessary complications.92 Health issues such as obesity, smoking, diabetes, depression, and other comorbidities are
prevalent in the MS patient community and must be addressed as part of comprehensive
care.93
In addition, nonadherence to therapy, discontinuation of therapy, and polypharmacy are key
unresolved issues in MS.94 It is common for patients with MS to feel overwhelmed by difficult
decisions and choices, and many struggle with the question of whether they are receiving the
best care for their condition.95-97 Optimal care of patients with MS requires an individualized,
patient-centered approach that:95
- Recognizes and treats multiple aspects of the person’s health;
- Addresses symptoms, systemic effects of the disease, and comorbidities;
- Adapts to the necessary changes throughout the person’s lifetime; and
- Encourages collaboration and communication within the healthcare team and across healthcare sectors.
Some of the key components for counseling patients with MS include:
- Setting realistic expectations for therapy. This may mean explaining to patients and family members that treatment will not make their existing MS symptoms disappear, but is a preventive step to minimize further damage from the disease process.
- Teaching patients to administer medications correctly (including injection technique when applicable.) Many manufacturers provide support services from trained nurses who work directly with the patient, often in the home setting, to give injection training.
- Proper medication storage, including refrigeration when applicable
- Management of potential AEs
- Instructions for following up with recommended monitoring
- Symptom management, comorbidities (e.g. bowel/bladder control)
- Assistance with coordinating care among health care providers
- Referral to peer support, support organizations
Tables 8 and 9 provide listings of MS support organizations and contact information, including
manufacturer-provided financial assistance and patient support programs.
| Table 8. National/International MS Organizations |
| Organization |
About |
Contact Information |
| National Multiple Sclerosis Society (NMSS) |
The largest MS advocacy group in the U.S., with the vision of “A World Free of MS.” |
nationalmssociety.org
contactusnmss@nmss.org
(800) 344-4867 |
| Multiple Sclerosis Association of America (MSAA) |
A national nonprofit organization dedicated to improving lives through vital services and support. |
General Information email:
webmaster@mymsaa.org
MS questions email:
MSquestions@mymsaa.org
Phone: (856) 488-4500
Helpline: (800)-532-7667 ext. 154 |
| Multiple Sclerosis Foundation (MSF) |
A national nonprofit organization offering support groups and grant-based assistance. |
Email: support@msfocus.org
Phone: (888) 673-6287 |
| North American Research Committee on Multiple Sclerosis (NARCOMS) |
An international patient registry service to collect information about living with MS that can be utilized by researchers to further the advancement of MS care. |
Email: MSregistry@narcoms.org
Phone: (800) 253-7884 |
| Consortium of Multiple Sclerosis Centers (CMSC) |
An organization of MS care professionals and centers focused on education of health care team and support programs to benefit people with MS. |
www.MScare.org
Email: info@MScare.org
Phone: (201) 487-1050 |
| Table 9. Pharmaceutical Company-Sponsored Nurse and Care Support Programs |
| Service |
About |
Contact Information |
MS Active Source
(Biogen)
Avonex, Plegridy, Tecfidera, Tysabri |
In-person injection training services; in home follow-up visits; phone counseling available 24/7. |
800-456-2255
www.msactivesource.com |
Beta Plus Program
(Bayer)
Betaseron |
Phone counseling available 24/7; individual injection training, in-person nurse visits. |
800-788-1467
www.betaseron.com |
MS Lifelines
(EMD Serono)
Rebif |
Call center available 27/7; Nurse support available 7 days a week (till 10 pm ET weekdays and till 5 pm ET weekends). In-home injection training services. |
877-447-3243
www.mslifelines.com |
Shared Solutions
(Teva)
Copaxone |
Call center M–F 8 am to 8 pm Central Time; after-hours nursing support available if needed. In-person injection training, refresher training, nurse support. |
800-887-8100
www.copaxone.com |
MS One to One
(Genzyme)
Aubagio |
24/7 access to on-call nurse; regularly appointed nurse available during business hours; no in-person visits. |
855-676-6326
www.msOnetoOne.com |
Go Program
Novartis
(Extavia, Gilenya) |
Call center available 8 am – 9 pm Eastern M–F. Nurse support for injection training and case management. |
866-398-2842 (Extavia)
www.extavia.com
800-445-3992 (Gilenya)
www.gilenya.com |
SUMMARY
The current management of MS involves expanded roles for pharmacists in many practice
settings. Diagnosis of MS is on the increase, more therapeutic options are available, and yearly
costs of therapy continue to rise. Pharmacists—especially those involved in formulary decision-making and specialty pharmacy—need updated information and greater clinical competency in
MS management.
We know that DMT offers significant clinical benefits for patients for MS in terms of delayed
onset of progressive disease, reduced relapse rates, and even survival benefits. Response to MS
therapy is highly individualized, so there are challenges when trying to rank drugs by efficacy
and cost. "Stronger" is not necessarily better for every patient, nor is "cheaper" always the
most cost-effective approach. Instead, therapy should be personalized for the individual and
monitored closely. Pharmacists need to maintain updated information about MS agents, as the
available list of treatment options changes regularly. Patients' needs include help with risk-benefit analysis; knowledge of dosage, administration, and monitoring; and financial assistance
programs to offset costs, including copayment costs.
REFERENCES
- Habibi M, Kuttab HM. Management of multiple sclerosis and the integration of related specialty pharmacy programs within health systems. Am J Health Syst Pharm. 2016;73(11):811-819.
- Hanson RL, Habibi M, Khamo N, Abdou S, Stubbings J. Integrated clinical and specialty pharmacy practice model for management of patients with multiple sclerosis. Am J Health Syst Pharm. 2014;71(6):463-469.
- Miller RM, Happe LE, Meyer KL, Spear RJ. Approaches to the management of agents used for the treatment of multiple sclerosis: consensus statements from a panel of U.S. managed care pharmacists and physicians. J Manag Care Pharm. 2012;18(1):54-62.
- Carlin CS, Higuera L, Anderson S. Improving Patient-Centered Care by Assessing Patient Preferences for Multiple Sclerosis Disease-Modifying Agents: A Stated-Choice Experiment. Perm J. 2017;21.
- Cofield SS, Thomas N, Tyry T, Fox RJ, Salter A. Shared Decision Making and Autonomy Among US Participants with Multiple Sclerosis in the NARCOMS Registry. Int J MS Care. 2017;19(6):303-312.
- Bandari DS, Sternaman D, Chan T, Prostko CR, Sapir T. Evaluating risks, costs, and benefits of new and emerging therapies to optimize outcomes in multiple sclerosis. J Manag Care Pharm. 2012;18(9):1-17.
- Goodin DS. The epidemiology of multiple sclerosis: insights to disease pathogenesis. Handbook of Clinical Neurology. 2014;122:231-266.
- Bjartmar C, Trapp BD. Axonal and neuronal degeneration in multiple sclerosis: mechanisms and functional consequences. Curr Opin Neurol. 2001;14(3):271-278.
- Hirst C, Ingram G, Pearson O, Pickersgill T, Scolding N, Robertson N. Contribution of relapses to disability in multiple sclerosis. J Neurol. 2008;255(2):280-287.
- Lublin FD. The incomplete nature of multiple sclerosis relapse resolution. J Neurol Sci. 2007;256 Suppl 1:S14-18.
- Lublin FD, Baier M, Cutter G. Effect of relapses on development of residual deficit in multiple sclerosis. Neurology. 2003;61(11):1528-1532.
- Lublin FD, Reingold SC, Cohen JA, et al. Defining the clinical course of multiple sclerosis: the 2013 revisions. Neurology. 2014;83(3):278-286.
- National Multiple Sclerosis Society. Primary progressive MS (PPMS). http://www.nationalmssociety.org/What-is-MS/Types-of-MS/Primary-progressive-MS.
- National MS Society. Secondary progressive MS. Available at: NationalMSsociety.org.
- National Multiple Sclerosis Society. Medications. Available at: https://www.nationalmssociety.org/Treating-MS/Medications.
- Le Page E, Leray E, Edan G. Long-term safety profile of mitoxantrone in a French cohort of 802 multiple sclerosis patients: a 5-year prospective study. Mult Scler. 2011;17(7):867-875.
- National Multiple Sclerosis Society (NMSS). FDA Approves Two New Generic Forms of Copaxone® (Glatiramer Acetate). October 4, 2017. Updated 10/6/2017. Available at: https://www.nationalmssociety.org/About-the-Society/News/FDA-Approves-Two-New-Generic-Forms-of-Copaxone®-(G.
- U.S. Food & Drug Administration. Zinbryta (daclizumab) Injection. Drug Approvals and Databases. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/761029Orig1s000TOC.cfm.
- Munsell M, Frean M, Menzin J, Phillips AL. An evaluation of adherence in patients with multiple sclerosis newly initiating treatment with a self-injectable or an oral disease-modifying drug. Patient Prefer Adherence. 2017;11:55-62.
- Johnson KM, Zhou H, Lin F, Ko JJ, Herrera V. Real-World Adherence and Persistence to Oral Disease-Modifying Therapies in Multiple Sclerosis Patients Over 1 Year. J Manag Care Spec Pharm. 2017;23(8):844-852.
- Alroughani R, Altintas A, Al Jumah M, et al. Pregnancy and the use of disease-modifying therapies in patients with multiple sclerosis: benefits versus risks. Mult Scler Int. 2016;2016:1034912.
- McGinley MP, Moss BP, Cohen JA. Safety of monoclonal antibodies for the treatment of multiple sclerosis. Expert Opin Drug Saf. 2017;16(1):89-100.
- Coles AJ, Cohen JA, Fox EJ, et al. Alemtuzumab CARE-MS II 5-year follow-up: Efficacy and safety findings. Neurology. 2017;89(11):1117-1126.
- U.S. Food & Drug Administration. Ocrevus (ocrelizumab) injection. Drug Approvals and Databases. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2017/761053Orig1s000TOC.cfm.
- Radick L, Mehr SR. The Latest Innovations in the Drug Pipeline for Multiple Sclerosis. Am Health Drug Benefits. 2015;8(8):448-453.
- Giovannoni G. Cladribine to Treat Relapsing Forms of Multiple Sclerosis. Neurotherapeutics. 2017;14(4):874-887.
- Giovannoni G, Soelberg Sorensen P, Cook S, et al. Safety and efficacy of cladribine tablets in patients with relapsing-remitting multiple sclerosis: Results from the randomized extension trial of the CLARITY study. Mult Scler. 2017:1352458517727603.
- Gajofatto A. Spotlight on siponimod and its potential in the treatment of secondary progressive multiple sclerosis: the evidence to date. Drug Des Devel Ther. 2017;11:3153-3157.
- Moreno Torres ID, Garcia-Merino A. Anti-CD20 monoclonal antibodies in multiple sclerosis. Expert Rev Neurother. 2016.
- Freedman MS, Hughes B, Mikol DD, et al. Efficacy of disease-modifying therapies in relapsing remitting multiple sclerosis: a systematic comparison. Eur Neurol. 2008;60(1):1-11.
- Kieseier BC, Wiendl H, Hartung HP, Leussink VI, Stuve O. Risks and benefits of multiple sclerosis therapies: need for continual assessment? Curr Opin Neurol. 2011;24(3):238-243.
- Weber MS, Menge T, Lehmann-Horn K, et al. Current treatment strategies for multiple sclerosis -efficacy versus neurological adverse effects. Curr Pharm Des. 2012;18(2):209-219.
- Goodin DS, Traboulsee A, Knappertz V, et al. Relationship between early clinical characteristics and long term disability outcomes: 16 year cohort study (follow-up) of the pivotal interferon beta-1b trial in multiple sclerosis. J Neurol Neurosurg Psychiatry. 2012;83(3):282-287.
- Rotstein DL, Healy BC, Malik MT, Chitnis T, Weiner HL. Evaluation of no evidence of disease activity in a 7-year longitudinal multiple sclerosis cohort. JAMA Neurol. 2015;72(2):152-158.
- Kappos L, De Stefano N, Freedman MS, et al. Inclusion of brain volume loss in a revised measure of 'no evidence of disease activity' (NEDA-4) in relapsing-remitting multiple sclerosis. Mult Scler. 2016;22(10):1297-1305.
- Giovannoni G, Turner B, Gnanapavan S, Offiah C, Schmierer K, Marta M. Is it time to target no evident disease activity (NEDA) in multiple sclerosis? Mult Scler Relat Disord. 2015;4(4):329-333.
- Hincapie AL, Penm J, Burns CF. Factors Associated with Patient Preferences for Disease-Modifying Therapies in Multiple Sclerosis. J Manag Care Spec Pharm. 2017;23(8):822-830.
- Freedman MS, Rush CA. Severe, Highly Active, or Aggressive Multiple Sclerosis. Continuum (Minneap Minn). 2016;22(3):761-784.
- Rush CA, MacLean HJ, Freedman MS. Aggressive multiple sclerosis: proposed definition and treatment algorithm. Nat Rev Neurol. 2015;11(7):379-389.
- Soelberg Sorensen P. Safety concerns and risk management of multiple sclerosis therapies. Acta Neurol Scand. 2017;136(3):168-186.
- Rieckmann P, Boyko A, Centonze D, et al. Achieving patient engagement in multiple sclerosis: A perspective from the multiple sclerosis in the 21st Century Steering Group. Mult Scler Relat Disord. 2015;4(3):202-218.
- Rae-Grant A, Day G, Marrie RA, et al. Evidence-based practice guideline: disease-modifying therapies for multiple sclerosis: report of the Guideline Development, Dissemination, and Implementation Subcommittee (GDDI) of the American Academy of Neurology. https://http//www.aan.com/uploadedFiles/Website_Library_Assets/Documents/2.Clinical_ Guidelines/3.Browse_By_Status/4.Guidelines_Under_Development/MS DMT guideline manuscript for public comment 2016-09-09.pdf.
- Otero-Romero S, Amato MP, Chandraratna D, et al. ECTRIMS-EAN clinical practice guideline on pharmacological management of multiple sclerosis. Presented at ECTRiMS, Paris, France. Sept 16, 2017. Abstract 255.
- Milo R. Therapeutic strategies targeting B-cells in multiple sclerosis. Autoimmun Rev. 2016.
- Kretzschmar B, Pellkofer H, Weber MS. The Use of Oral Disease-Modifying Therapies in Multiple Sclerosis. Curr Neurol Neurosci Rep. 2016;16(4):38.
- Das RK, Brar SK, Verma M. Recent advances in the biomedical applications of fumaric acid and its ester derivatives: The multifaceted alternative therapeutics. Pharmacol Rep. 2016;68(2):404-414.
- Korsen M, Bragado Alonso S, Peix L, Broker BM, Dressel A. Cladribine Exposure Results in a Sustained Modulation of the Cytokine Response in Human Peripheral Blood Mononuclear Cells. PLoS One. 2015;10(6):e0129182.
- Klotz L, Wiendl H. Monoclonal antibodies in neuroinflammatory diseases. Expert Opin Biol Ther. 2013;13(6):831-846.
- Institute for Clinical and Economic Review (ICER). 2017 Final Evidence Report – DMTs for RRMS and PPMS. Available at: https://icer-review.org/wpcontent/uploads/.../ICER_Draft_MS_Report_112216.pdf.
- Carroll WM. Clinical trials of multiple sclerosis therapies: improvements to demonstrate long-term patient benefit. Mult Scler. 2009;15(8):951-958.
- Coyle PK. Early treatment of multiple sclerosis to prevent neurologic damage. Neurology. 2008;71(24 Suppl 3):S3-7.
- Ford C, Goodman AD, Johnson K, et al. Continuous long-term immunomodulatory therapy in relapsing multiple sclerosis: results from the 15-year analysis of the US prospective open-label study of glatiramer acetate. Mult Scler. 2010;16(3):342-350.
- Tremlett H, Yousefi M, Devonshire V, Rieckmann P, Zhao Y, Neurologists UBC. Impact of multiple sclerosis relapses on progression diminishes with time. Neurology. 2009;73(20):1616-1623.
- Goodin DS, Reder AT, Ebers GC, et al. Survival in MS: a randomized cohort study 21 years after the start of the pivotal IFNbeta-1b trial. Neurology. 2012;78(17):1315-1322.
- Reder AT, Ebers GC, Traboulsee A, et al. Cross-sectional study assessing long-term safety of interferon-beta-1b for relapsing-remitting MS. Neurology. 2010;74(23):1877-1885.
- Mendes D, Alves C, Batel-Marques F. Benefit-Risk of Therapies for Relapsing-Remitting Multiple Sclerosis: Testing the Number Needed to Treat to Benefit (NNTB), Number Needed to Treat to Harm (NNTH) and the Likelihood to be Helped or Harmed (LHH): A Systematic Review and Meta-Analysis. CNS Drugs. 2016;30(10):909-929.
- Soelberg Sorensen P. Safety concerns and risk management of multiple sclerosis therapies. Acta Neurol Scand. 2016.
- McGinley MP, Moss BP, Cohen JA. Safety of monoclonal antibodies for the treatment of multiple sclerosis. Expert Opin Drug Saf. 2016:1-12.
- Bruck W, Gold R, Lund BT, et al. Therapeutic decisions in multiple sclerosis: moving beyond efficacy. JAMA Neurol. 2013;70(10):1315-1324.
- Miller AE. Switching or discontinuing disease-modifying therapies for multiple sclerosis. Continuum (Minneap Minn). 2016;22(3, Multiple Sclerosis and Other Demyelinating Diseases):851-863.
- Raju GK, Gurumurthi K, Domike R. Benefit-Risk Analysis for Decision-Making: An Approach. Clin Pharmacol Ther. 2016.
- Food and Drug Administration (FDA). Supplemental NDA Approval to Release REMS Requirement. Available at: https://http//www.accessdata.fda.gov/drugsatfda_docs/appletter/2016/022527Orig1s021l tr.pdf.
- Berger JR, Houff S. Opportunistic infections and other risks with newer multiple sclerosis therapies. Ann Neurol. 2009;65(4):367-377.
- Lebrun C, Debouverie M, Vermersch P, et al. Cancer risk and impact of disease-modifying treatments in patients with multiple sclerosis. Mult Scler. 2008;14(3):399-405.
- Robinson CL, Guo M. Fingolimod (Gilenya) and melanoma. BMJ Case Rep. 2016;2016.
- Buttmann M, Seuffert L, Mader U, Toyka KV. Malignancies after mitoxantrone for multiple sclerosis: A retrospective cohort study. Neurology. 2016;86(23):2203-2207.
- Lemtrada (alemtuzumab) [package insert]. Cambridge, MA: Genzyme. 2017.
- Auricchio F, Scavone C, Cimmaruta D, et al. Drugs approved for the treatment of multiple sclerosis: review of their safety profile. Expert Opin Drug Saf. 2017;16(12):1359-1371.
- Biogen. TYSABRI (natalizumab): PML in Patients Receiving TYSABRI (USA TYSABRI PML Update). Updated September 2017.
- Berger JR. Classifying PML risk with disease modifying therapies. Mult Scler Relat Disord. 2017;12:59-63.
- Coles AJ, Boyko AN, Cohen JA, et al. Alemtuzumab provides durable improvements in clinical outcomes in treatment-naive patients with active relapsing-remitting multiple sclerosis over 6 years in the absence of continuous treatment (CARE-MS I). European Committee for Treatment Research in Multiple Sclerosis (ECTRIMS) 2016. London, England. Abstract 213.
- Fox EJ, Alroughani R, Brassat D, et al. Efficacy of alemtuzumab is durable over 6 years in patients with active relapsing-remitting multiple sclerosis and an inadequate response to prior therapy in the absence of continuous treatment (CARE-MS II). European Committee for Treatment Research in Multiple Sclerosis (ECTRIMS) 2016. London, England. Abstract P1150.
- Meyer-Massetti C, Cheng CM, Schwappach DL, et al. Systematic review of medication safety assessment methods. Am J Health Syst Pharm. 2011;68(3):227-240.
- Chisholm-Burns MA, Kim Lee J, Spivey CA, et al. US pharmacists' effect as team members on patient care: systematic review and meta-analyses. Med Care. 2010;48(10):923-933.
- Coyle PK. Switching therapies in multiple sclerosis. CNS Drugs. 2013;27(4):239-247.
- Coyle PK. Switching algorithms: from one immunomodulatory agent to another. J Neurol. 2008;255 Suppl 1:44-50.
- Reynolds MW, Stephen R, Seaman C, Rajagopalan K. Healthcare resource utilization following switch or discontinuation in multiple sclerosis patients on disease modifying drugs. J Med Econ. 2010;13(1):90-98.
- American Academy of Neurology (AAN). Position Statement. Availability of DMTs for Treatment of Relapsing Forms of MS. 2015. Available at: www.aan.com.
- NARCOMS MS in America Study 2014. Special Report: Oral Treatments for MS. Available at: www.multiplesclerosis.net/special-report-2014-oral-treatments-for-ms/switching-to-oralmedications
- Costello K, Kennedy P, Scanzillo J. Recognizing nonadherence in patients with multiple sclerosis and maintaining treatment adherence in the long term. Medscape J Med. 2008;10(9):225.
- Rio J, Nos C, Tintore M, et al. Defining the response to interferon-beta in relapsing-remitting multiple sclerosis patients. Ann Neurol. 2006;59(2):344-352.
- Sormani MP, Bruzzi P. MRI lesions as a surrogate for relapses in multiple sclerosis: a meta-analysis of randomised trials. Lancet Neurol. 2013;12(7):669-676.
- Bandari DS, Sternaman D, Chan T, Prostko CR, Sapir T. Evaluating risks, costs, and benefits of new and emerging therapies to optimize outcomes in multiple sclerosis. J Manag Care Pharm. 2012;18(9):1-17.
- Waubant E. Overview of treatment options in multiple sclerosis. J Clin Psychiatry. 2012;73(6):e22.
- Plosker GL. Interferon-beta-1b: a review of its use in multiple sclerosis. CNS Drugs. 2011;25(1):67-88.
- Hartung DM, Bourdette DN, Ahmed SM, Whitham RH. The cost of multiple sclerosis drugs in the US and the pharmaceutical industry: Too big to fail? Neurology. 2015;84(21):2185-2192.
- MS Coalition. The Use of Disease-Modifying Therapies in Multiple Sclerosis: Principles and Current Evidence. 2016. Available at: http://www.nationalmssociety.org/getmedia/5ca284d3-fc7c-4ba5-b005-ab537d495c3c/DMT_Consensus_MS_Coalition_color.
- Express Scripts 2016 Drug Trend Report. Available at: Available at: http://lab.express-scripts.com/lab/drug-trend-report/commercial-drug-trend-report.
- Express Scripts. Financial Assistance Programs Positively Impact Adherence. Nov 29, 2016. Available at: http://lab.express-scripts.com/lab/insights/adherence/patient-assistance-programspositively-impact-adherence.
- Clavelou P. Can we optimize our teams? Multidisciplinary care for multiple sclerosis. Exp Rev Neurotherapeutics. 2013;13(12 Suppl):39-44.
- Rieckmann P, Boyko A, Centonze D, et al. Future MS care: a consensus statement of the MS in the 21st Century Steering Group. J Neurol. 2013;260(2):462-469.
- Ziemssen T, Derfuss T, de Stefano N, et al. Optimizing treatment success in multiple sclerosis. J Neurol. 2016;263(6):1053-1065.
- Salter AR, Marrie RA, Agashivala N, et al. Patient perspectives on switching disease-modifying therapies in the NARCOMS registry. Patient Prefer Adherence. 2014;8:971-979.
- Rieckmann P, Boyko A, Centonze D, et al. Achieving patient engagement in multiple sclerosis: A perspective from the multiple sclerosis in the 21st Century Steering Group. Mult Scler Relat Disord. 2015;4(3):202-218.
- Derfuss T. Personalized medicine in multiple sclerosis: hope or reality? BMC Med. 2012;10:116.
- De Jager PL. Identifying patient subtypes in multiple sclerosis and tailoring immunotherapy: challenges for the future. Ther Adv Neurol Disord. 2009;2(6):8-19.
- Miller A, Avidan N, Tzunz-Henig N, et al. Translation towards personalized medicine in Multiple Sclerosis. J Neurol Sci. 2008;274(1-2):68-75.
Back Top