
Expired activity
Please go to the PowerPak
homepage and select a course.
Beyond Fiber and Laxatives: Advising Patients with Chronic and Refractory Constipation - Article
INTRODUCTION
Constipation is described as difficulty passing stool or infrequent bowel movements.1 This may be accompanied by symptoms such as straining or the feeling of incomplete evacuation. Constipation is one of the most common gastrointestinal (GI) disorders, affecting an estimated 14% of the population. Women have been reported to be twice as likely to experience constipation compared with men. Older age and lower socioeconomic status have also been associated with a higher risk of developing constipation. Constipation can compromise patients' quality of life as it interferes with normal daily function and negatively affects well-being. Even so, fewer than half of patients suffering symptoms of constipation on an ongoing basis seek support and advice from a healthcare provider.1
Many comorbid conditions, medications, or lifestyle choices can cause or contribute to development of constipation.2 However, constipation does not have an organic cause in many patients. Chronic constipation that is not a result of an organic medical issue is considered a functional bowel disorder (FBD), and can further be classified as either functional constipation (FC) or constipation-predominant irritable bowel syndrome (IBS-C).3 The Rome IV criteria provide diagnostic criteria for various FBDs.3
DIAGNOSTIC CONSIDERATIONS
Functional Constipation
Functional constipation is a bowel disorder defined by difficult, infrequent, or incomplete defecation in the absence of alarm symptoms or secondary causes.3 Patients may report associated abdominal pain or bloating, but these are not defining symptoms of FC. If abdominal pain and bloating predominate, the patient may meet criteria for IBS-C instead of FC. FC is considered to be a chronic bowel disorder; to meet diagnostic criteria, symptom onset should occur 6 months or more before diagnosis, and symptoms should be present during the past 3 months. Because of the ongoing nature of FC, it is often referred to as chronic idiopathic constipation (CIC).
Refractory constipation (RC) is a commonly used term that the Rome IV criteria do not define. In an attempt to define refractory constipation, Soh and colleagues completed a systematic review of studies evaluating patients with refractory or intractable constipation.4 The authors found that studies did not consistently report details of previous therapies when describing patients with refractory constipation. Even so, many patients in the systematic review received invasive surgical therapy, including colectomy, to treat their constipation. The authors suggest that ongoing symptoms of constipation following 12 weeks of continuous treatment with an agent or a combination of agents at doses shown to be effective for constipation constitutes a suitable trial to classify the patient as having pharmacologically refractory constipation. Patients who are classified as having refractory constipation may benefit from escalation of therapy, combination therapy, as well as referral for additional diagnostic testing and treatment by a gastroenterologist.
Constipation-Predominant Irritable Bowel Syndrome
Irritable bowel syndrome (IBS) is a type of FBD.3 Its hallmark symptoms are chronic, recurrent abdominal pain or discomfort accompanied by a change in bowel habits without alarm symptoms or an underlying organic cause. IBS is classified based on a patient's predominant stool pattern as either constipation predominant (IBS-C), diarrhea predominant (IBS-D), mixed type (IBS-M), or unclassified type (IBS-U). Similar to FC, to fulfill Rome IV diagnostic criteria for IBS, symptoms must have started at least 6 months prior to diagnosis, and should be present for at least the past 3 months.
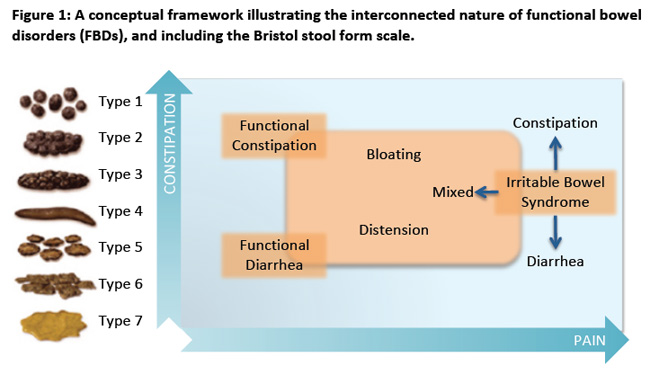
The prevalence of IBS is estimated to be between 10% and 15% with women affected twice as often as men.5 IBS diagnosis peaks in early adulthood; however, it may be diagnosed at any age. It is reported to be the most commonly diagnosed GI disorder and constitutes up to half of all referrals to gastroenterologists.5 Details regarding the Rome IV diagnostic criteria for FC and IBS-C can be found in Box 1. While the individual FBDs each have distinct diagnostic criteria, it is important to recognize that they exist on a continuum of bowel disorders, and many of the same treatments are used for both FC and IBS-C (Figure 1).
Box 1. Rome IV diagnostic criteria for functional constipation (FC) and constipation- predominant irritable bowel syndrome (IBS-C)3,5,6
FC
- Symptom onset at least 6 months before diagnosis, with symptoms present for at least the past 3 months.
- Two or more of the following for at least 25% of defecations:
- Straining
- Lumpy or hard stools
- Sensation of incomplete evacuation
- Sensation of anorectal obstruction/blockage
- Manual maneuvers to facilitate defecation
- Fewer than 3 spontaneous bowel movements per week
- Loose stools are rarely present without the use of laxatives
- IBS diagnostic criteria are not met
IBS
- Symptom onset at least 6 months before diagnosis, with symptoms present at least 1 day per week for at least the past 3 months.
- Recurrent abdominal pain, on average, at least 1 day per week in the last 3 months, associated with 2 or more of the following criteria:
- Related to defecation
- Associated with a change in frequency of stool
- Associated with a change in form or appearance of stool
- To be classified as IBS-C, more than 25% of bowel movements must be classified as Bristol stool form types 1 or 2 (constipation) and fewer than 25% must be classified as Bristol stool form types 6 or 7 (diarrhea)
|
| Abbreviations: FC, functional constipation; IBS, irritable bowel syndrome; IBS-C, constipation- predominant irritable bowel syndrome. |
PATIENT CHALLENGES IN MANAGING CONSTIPATION
Many patients experience chronic constipation, whether it is CIC or IBS-C; however, fewer than half of patients with chronic symptoms seek the advice of a healthcare provider.1 Patients' reasons for not reporting constipation symptoms to a healthcare provider may vary. Patients may not discuss their constipation with a healthcare provider because they may not appreciate the severity of their symptoms, or they may lack confidence in describing their symptom severity to their healthcare provider.
A pharmacist is one of the most easily accessible healthcare providers, and is also available for consultation regarding many over-the-counter (OTC; nonprescription) laxatives that patients with constipation symptoms often seek to use. Even so, in a study of 500 patients who purchased OTC laxatives, only 78 (16%) reported consulting with a pharmacist when making their selection, while 361 (72%) reported a desire to receive information or advice regarding laxative selection from a pharmacist.8
To facilitate discussions about constipation, which may be a sensitive topic for some patients, pharmacists are encouraged to use a tool such as the Bristol Stool Form Scale (BSFS; Figure 1).9 The BSFS is a 7-point scale wherein a score of 1 or 2 indicates constipation, 3 or 4 indicate normal stool formation, and 5, 6, or 7 indicate diarrhea. The scale provides the healthcare provider and the patient with a common concrete set of terms to use to describe bowel movements. This validated scale has been shown to highly correlate with GI transit time, even more so than the frequency of bowel movements.
Additional reasons patients may choose not to seek the advice of a healthcare provider include embarrassment, feeling that the healthcare providers respond with a lack of empathy, and feeling that the healthcare provider would not offer helpful recommendations.10 This may lead patients to self-treat their symptoms with OTC medications, or with herbal remedies or complementary and alternative medicine.
Patients with CIC or IBS-C may not experience relief of their constipation with OTC treatments, or they may experience adverse effects that limit the utility of these therapies. These hurdles can be overcome by healthcare providers who adopt patient-centered approaches to care such as motivational interviewing,11 in which the healthcare provider listens without judgment while empowering the patient to be a partner in their healthcare decision-making process.
Another approach, which is popular in the United Kingdom, is the WWHAM process, an acronym for structured patient communication using the following questions12:
- Who is the patient
- What are the symptoms
- How long have the symptoms been present
- Action taken
- Medication taken
Using this type of structured approach ensures that the healthcare provider is taking an adequate history, fully listening to the patient's concerns, and responding with an appropriate course of action, whether it is offering advice regarding self-care OTC treatment options, or referral for additional diagnostic testing or prescription therapy. These structured approaches can also aid in determining whether a patient is experiencing treatment failure and requires further evaluation or escalation of their therapy.
PATHOLOGY OF CONSTIPATION
Constipation can occur secondary to a number of causes (Table 1).2,6,13 A thorough patient history can help in ruling out secondary causes, as well as confirm the absence of alarm features (positive family history of colorectal cancer, rectal bleeding in the absence of documented bleeding hemorrhoids or anal fissures, unintentional weight loss, or anemia).3 Once secondary causes and alarm features are ruled out, patients with ongoing constipation may be classified as having a FBD — either CIC or IBS-C, depending on which criteria are fulfilled.
| Table 1. Common Causes of Secondary Constipation2,6,13 |
| Medications |
Metabolic Disorders |
Neuropathy or Neurologic Disorders |
Other Conditions |
Opioids
Diuretics
Calcium channel
blockers
Antidepressants
Antihistamines
Antipsychotics
Antispasmodics
Antiparkinson agents
Metallic ions (iron,
calcium)
Antacids containing
aluminum, calcium
Nonsteroidal anti-
inflammatory drugs |
Diabetes
Hypothyroidism
Hyperparathyroidism
Chronic kidney disease
Electrolyte imbalance
– Hypercalcemia
– Hypokalemia
– Hypomagnesemia |
Autonomic neuropathy
Hirchsprung disease
Amyloidosis
CNS lesions
Cognitive impairment
Parkinson disease
Multiple sclerosis
Spinal cord injury
Stroke |
Eating disorders
Diet
– Low fiber
– Low caloric intake
– High protein
Colonic obstruction or pseudo-obstruction |
Three clinical subgroups of constipation have been defined by the American Gastroenterological Association (AGA): those with normal transit constipation, slow transit constipation, and defecatory disorders or outlet dysfunction.14 Evaluation of colonic transit and anorectal function allows constipation to be classified within these subgroups, and this classification may aid in tailoring therapy. Normal colonic transit time is typically between 20 and 72 hours; normal transit constipation is the most common type of constipation, especially in patients with IBS-C or CIC.6 A slower-than-normal colonic transit time, usually 5 or more days, is considered slow transit constipation.6 This type of constipation is often the result of slow colon peristalsis secondary to smooth muscle or neural dysfunction. Opioids are a common cause of slow transit constipation and should be ruled out as a cause in suspected CIC or IBS-C. Opioid-induced constipation (OIC) is now a separate FBD defined by the Rome IV criteria. The treatment approaches employed in OIC differ from those described in this program.
There is some evidence that FC occurs in families, although genetic links have not been definitively demonstrated.3 Limited evidence shows pediatric lifestyle habits (e.g., low fiber diet, reduced fluid intake, and ignoring the urge to defecate) may be correlated with constipation in adulthood.3 Additional data support a reduction in constipation when a high- fiber diet is consumed, when hydration is maintained, and when patients engage in physical activity. Patients may be at risk for IBS-C based on the interplay of genetic, environmental, or psychosocial factors.3,13 IBS may be triggered following gastroenteritis or result from food intolerances, chronic stress, diverticulitis, or surgery. These may result in alterations in GI motility and colonic transit, visceral hypersensitivity, GI immune activation, or alterations in the colonic microbiome or brain-gut function.
Assessing Constipation
When assessing a patient with constipation, a thorough history is of utmost importance. The history should include specifics regarding the onset and duration of symptoms, frequency of bowel movements, additional symptoms such as abdominal pain, as well as the classification of stool form using a validated tool such as the BSFS that offers a noninvasive approach to assessing for normal or slow colonic transit.9 Inquiring about specific symptoms can help differentiate CIC from IBS-C, and provides the healthcare provider with an opportunity to offer the patient education regarding constipation and, later, to tailor therapy to the symptoms the patient is experiencing. In a survey of 557 patients with chronic constipation, the most commonly reported symptoms were straining (79%), hard stools (71%), abdominal discomfort (62%), bloating (57%), infrequent bowel movements (57%), and feelings of incomplete evacuation (54%).15
Additional discussion regarding comorbidities; current prescription and OTC medications; supplements, vitamins, or herbals being used; diet; and lifestyle including type and amount of physical activity should be undertaken, as these factors can cause or exacerbate constipation.3,6,13 Alarm features should be discussed to ensure these are not present as well. If alarm features are present, or patients do not fit into a FBD category, referral to a primary care provider or gastroenterologist is required for additional objective tests such as evaluations of colonic transit or anorectal function.3
TREATMENT OF CIC AND IBS-C
Nonpharmacologic Treatment
Nonpharmacologic approaches should be initiated in all patients with constipation, either alone or in combination with pharmacologic therapy. Patients should be encouraged to increase physical activity, especially in the early morning when colonic motility naturally increases.16 An increase in dietary fiber is often considered a mainstay of treatment of constipation.6,16 Patients should be counseled to gradually increase dietary fiber to 25 to 30 grams per day to avoid abdominal cramping and bloating that may occur with rapid titration of dietary fiber. In addition to increasing fiber intake, consuming 1.5 to 2 liters of fluid daily, especially in patients who may be dehydrated, can lessen the symptoms of constipation. In addition, drinking a hot caffeinated beverage after waking, and eating breakfast within 1 hour of waking may also alleviate symptoms of constipation. Impaired sleep can exacerbate constipation symptoms; ensuring adequate sleep through sleep hygiene techniques may be recommended.3
Toilet training is a lifestyle modification that can be easily implemented as well.6,17 This entails having a routine of toileting either in the morning or evening, even in the absence of urge. Patients should be encouraged not to ignore the urge to toilet, not to remain on the toilet for more than 10 minutes, to avoid straining, and to use deep relaxation techniques while toileting.
In addition, patients should employ a posture to allow for efficient toileting: sitting on the toilet leaning forward with feet supported on a short stool that positions the knees higher than the hips will help to straighten the anorectal angle.6
Patients with IBS-C may respond to dietary changes that address food intolerance or sensitivity or through alteration of the gut microbiome.18-20 Some 90% of patients with IBS restrict their diets to alter their GI symptoms.19
Gluten elimination is one of the dietary changes most frequently applied by patients with IBS.18,19 Before a patient with IBS institutes a gluten-free diet, it is advisable to test for celiac sprue to verify that, if IBS symptoms improve, the improvement is due to non-celiac wheat sensitivity. Patients with IBS who eliminate gluten from their diets have been shown to experience improvements in IBS symptoms as well as quality of life compared to baseline.19
Another dietary change that has received attention in the treatment of IBS is reduction of dietary fermentable oligosaccharides, disaccharides, monosaccharides, and polyols, or FODMAPs.18,19 FODMAPs are foods containing short-chain carbohydrates that are poorly absorbed from the small intestine. These carbohydrates pass into the colon where they are fermented by bacteria, creating luminal gas that results in bloating and distension. Presence of FODMAPs in the colon may also quantitatively and qualitatively alter the colonic microbiome. Some examples of FODMAPs are beans and lentils, lactose-containing dairy products, high- fructose corn syrup, and some artificial sweeteners.
While elimination diets may be helpful for controlling symptoms of IBS, it is important to ensure that patients are receiving adequate nutrition. Recent analyses indicate that elimination diets may result in a compromised nutritional status.20
OTC Approaches
Numerous treatment options for constipation are available without a prescription and used by patients to self-treat their symptoms. Patients with CIC or IBS-C may benefit from some of the available OTC options, whether they are used alone or in combination with prescription therapies.
Fiber supplementation, whether through dietary fiber intake or fiber products, is a reasonable first-line treatment option in those with CIC or IBS-C. It is generally most effective for patients with normal-transit constipation. Soluble fiber such as psyllium is generally better tolerated than insoluble wheat bran–based fiber, which is associated with higher rates of bloating, cramping, or abdominal distension. Patients should be counseled to slowly titrate the dose of fiber in their diet or supplement to avoid these adverse effects.
Fiber supplementation is effective for treatment of CIC with a reported number needed to treat (NNT) of 2.21 Fiber, especially soluble fiber, also appears to be effective in the treatment of IBS- C with an NNT of 7.22 Because of the need to titrate the initial dose of fiber and with differing bulk-forming mechanisms of fiber therapy, products' effects are seen gradually over days to weeks of treatment, and fiber therapy may be considered a long-term treatment. Patients using fiber therapy should also ensure that they drink adequate amounts of fluid to avoid worsening their constipation.
Docusate is a stool softener that acts as a surfactant, which can be used to promote softer stools. As with fiber use, adequate hydration is necessary for optimal efficacy of docusate.
Osmotic laxatives are products that mainly remain in the intestinal lumen where they draw fluid in to soften the stool. Electrolyte-based osmotic laxatives include magnesium-based products (magnesium hydroxide, magnesium citrate). These products tend to have a low associated risk of hypermagnesemia, but should be avoided in patients with kidney or cardiac disease. These products have a faster onset of action and can be used for acute treatment, although the data for their use in CIC and IBS-C are lacking.6 Sodium phosphate products are used in bowel preparation regimens and should not be used for CIC or IBS-C because of the risk of hyperphosphatemia or acute phosphate nephropathy.6 Lactulose, which is available by prescription only, and polyethelene glycol both are effective in the treatment of CIC.21 Polyethelene glycol has been shown to improve stool consistency and frequency IBS-C without improving abdominal pain or bloating.23
Stimulant laxatives such as bisacodyl and sennosides are recommended for short-term use, generally less than 4 weeks. With the availability of agents that are safe to use for longer-term therapy in CIC and IBS-C, stimulant laxatives are less preferred for treating chronic constipation symptoms.6 While OTC laxatives may be helpful in the treatment of CIC and IBS-C, they may not be sufficient in refractory cases, for which patients often require combination therapy with OTC and prescription treatments.24
Prescription Agents
Three intestinal secretagogues — lubiprostone, linaclotide, and plecanatide — are currently available by prescription only in the United States. All are approved by the U.S. Food and Drug Administration (FDA) for the treatment of CIC, and lubiprostone and linaclotide are also approved for the treatment of IBS-C. The secretagogues have been shown to be effective in both normal and slow transit constipation.
Table 2 lists available drug therapies for CIC and IBS-C, and Table 3 lists recommendations and quality of evidence of CIC and IBS-C treatments, as detailed in the following discussions of available prescription medications for these conditions.
| Table 2. Drug Therapy Options for the Treatment of CIC and IBS-C2,3,6,13,21,25,28,32 |
| Drugs Act |
Actions |
Dose |
Comments |
| Bulk-forming laxatives |
Soluble: Psyllium,
Inulin,
Methylcellulose,
Calcium polycarbophil
Insoluble:
Wheat bran |
Increases stool bulk
and fluid content
stimulating colonic
motility |
2.5 g one to three
times daily, up to 30
g/day in divided
doses |
Onset 12–72 hr; less effective in
slow transit constipation;
requires adequate water intake
to be effective; soluble forms
be incorporated into foods,
liquids, and recipes; safe in renal
and hepatic disease, pregnancy,
geriatrics; can cause gas and
bloating, titrate dose slowly |
| Stool softeners |
Docusate sodium
Docusate
potassium |
Acts as a surfactant to
reduce surface tension
of colonic content to
lubricate and soften
fecal matter |
50–400 mg daily |
Onset 1–3 days; requires
adequate water intake to be
effective |
| Osmotic laxatives |
Magnesium
hydroxide,
magnesium
citrate, sodium
phosphate
Lactulose
Polyethylene
glycol 3350 (PEG) |
Poorly absorbed ions
retain water in the
intestinal lumen.
Mainly reserved for
acute constipation or
bowel prep for
procedure.
Degraded by colonic
bacteria to carbon
dioxide, lactic acid, and
acetic acid. Causes fecal
acidification and
osmotic and
fermentative diarrhea.
Increases luminal fluid
via increasing osmotic
pressure |
Magnesium
hydroxide: 30–60
mL once daily or in
divided doses
15–30 mL once or
twice daily
17 g (1 heaping
tablespoonful of
powder) once or
twice daily in 8
ounces of water |
Fast onset (15 min to 3 h); avoid
in renal impairment, HF, cirrhosis;
FDA warning regarding sodium
phosphate and development of
acute phosphate nephropathy
Onset 1–2 days (may require
multiple doses); associated with
gas and bloating; syrup or powder
for solution may be more
palatable if mixed with fruit juice,
water, or milk.
Onset 1–3 days; safe in renal and
hepatic disease and pregnancy;
overall, well tolerated; may be
used longer term |
| Stimulant laxatives |
Bisacodyl
Sennosides |
Stimulates sensory
nerves in colonic
mucosa to inhibit water
resorption and
stimulate secretion/
motility of small
intestine and colon
Increases electrolytes
entering the bowel,
stimulates intestinal
motility |
10-15 mg by mouth
or 10-mg suppository
up to 3 times per
week
Dose range: 2 tablets
once daily to 4 tablets
twice daily |
Oral onset 6–12 h, suppository
within 1 h; oral tablets are
enteric coated. May cause gastric
or rectal irritation.
Tablets and liquid; onset 6–12 h;
can cause abdominal cramping,
electrolyte disturbances,
melanosis coli |
| Prosecretory age |
Lubiprostone
(Amitiza)
Linaclotide
(Linzess)
Plecanatide
(Trulance) |
Chloride channel (ClC-
2) activator; results
in intestinal fluid
secretion and increased
motility
Guanylate cyclase-C
agonist; increases
colonic fluid secretion
and accelerated transit
time
Guanylate cyclase-C
agonist; increases
colonic fluid secretion
and accelerated transit
time |
IBS-C: 8 mcg twice
daily
CIC: 24 mcg twice
daily
IBS-C: 290 mcg once
daily
CIC: 145 mcg once
daily or 72 mcg once
daily. Capsules may be
opened and sprinkled
on applesauce and
eaten, or mixed in
water to be swallowed
or placed in a feeding
tube.
IBS-C and CIC: 3 mg
tablet once daily.
May be crushed and
administered in
applesauce or water
or via feeding tube |
May reduce bloating and
abdominal pain; main adverse
effect is nausea; negative
pregnancy test required before
use
Take on empty stomach 30 min
before meal; common adverse
effects: diarrhea, abdominal pain,
flatulence, and abdominal
distension; contraindicated in
pediatric patients <6 years old and
in mechanical obstruction; avoid in
patients 6–17 years old;
pregnancy category C
Diarrhea is a common adverse
effect. Contraindicated in
pediatric patients <6 years old and
in mechanical obstruction; avoid in
patients 6–17 years old |
| Abbreviations used: CIC, chronic idiopathic constipation; CrCl, creatinine clearance; FDA, U.S. Food and Drug Administration; HF, heart failure; GI, gastrointestinal; IBS-C, constipation-predominant irritable bowel syndrome; MI, myocardial infarction. |
| Table 3. American College of Gastroenterology Recommendations and Quality of Evidence Regarding Treatments for CIC and IBS-C21,39 |
| Statement for CIC or IBS-C |
Recommendation/Quality of Evidence |
CIC
Fiber increases stool frequency
PEG increases stool frequency and consistence
Linaclotide is effective in CIC
Lubiprostone is effective
Lactulose increases stool frequency and consistency
Bisacodyl is effective
IBS-C
Fiber provides overall symptom relief but may cause bloating
and abdominal discomfort
No evidence that PEG improves overall symptoms and pain
Linaclotide is superior to placebo
Lubiprostone is superior to placebo |
Strong/low
Strong/high
Strong/high
Strong/high
Strong/low
Strong/moderate
Weak/moderate
Weak/low
Strong/high
Strong/moderate |
Lubiprostone
Lubiprostone is a prostaglandin E1 derivative and selective chloride channel-2 activator that exerts its effect in the small intestine to increase fluid secretion and gut motility. Lubiprostone has FDA-approved indications for the treatment of adults with CIC or OIC, and adult women with IBS-C.25
In a study of 124 patients with CIC who were randomized to receive lubiprostone 24 mcg twice daily or placebo for 4 weeks, those on lubiprostone had a significant increase in the number of spontaneous bowel movements (SBMs) per week in the first week of the trial. The lubiprostone group had a mean increase of 3.66 SBMs/week versus an increase of 1.26 SBMs/week in the placebo group (P <0.001). These findings were sustained at each weekly visit through week 4.26
Those investigators completed a second analysis of 201 patients who were given lubiprostone 24 mcg twice daily to treat CIC for up to 48 weeks. Changes in SBMs from baseline were seen in the first week of the study and were maintained throughout the 48 weeks P <0.0001). This was seen in patients with CIC (n = 167) as well as those who met diagnostic criteria for IBS-C (n = 34). Assessment of quality of life using the 36-item Short-Form Health Survey Questionnaire (SF-36) indicated that treatment with lubiprostone significantly improved the domains of physical functioning, bodily pain, general health, vitality, and role emotional at weeks 24 and 48 (P <0.05 for all domains at 24 and 48 weeks compared with baseline).26
Patients receiving lubiprostone reported more adverse effects than those receiving placebo (41.9% versus 16.1%, P = 0.005); all but one of the adverse effects in the study were reported to be mild. Diarrhea and nausea were reported most frequently in the lubiprostone group, with 9 patients (14.5%) reporting each of these within the first 4 weeks of treatment. In the longer study, 37.3% of patients reported diarrhea and 27.3% reported nausea after 48 weeks of lubiprostone.26
Lubiprostone 8 mcg twice daily performed well in a 12-week, randomized, double-blind, placebo-controlled trial of 1,171 patients with IBS-C. The primary outcome of overall response, which was defined as symptom relief compared with baseline as captured by a 7-point Likert scale in patients' electronic diaries, was achieved by 17.9% of patients with IBS compared with 10.1% of patients treated with placebo (P = 0.001). Patients receiving lubiprostone who achieved overall response also had significant improvements in scores for abdominal pain or discomfort, bloating, constipation severity, stool consistency, and straining (P <0.001 compared with nonresponders).27
Overall, half of all patients in the study reported an adverse effect, with 50% of patients in the lubiprostone group and 51% of patients receiving placebo reporting at least one adverse effect. The majority of adverse effects were mild or moderate in intensity. Nausea, diarrhea, and abdominal distention were most frequently reported (19% lubiprostone versus 14% placebo), and similar numbers of patients withdrew from the study because of adverse effects (9 patients taking lubiprostone and 12 patients on placebo). Study participants were able to reduce their lubiprostone dose to 8 mcg once daily if they experienced nausea or diarrhea for more than 2 days; the study did not report how many patients opted to reduce their dosage.27
Lubiprostone is generally well tolerated, with nausea being its most common adverse effect. A pooled analysis of clinical trial data of lubiprostone used in the treatment of CIC, OIC or IBS-C was published with the goal of characterizing the impact of nausea on the course of therapy of patients receiving lubiprostone.27 The authors found that patients treated with lubiprostone for CIC experienced nausea more commonly than those treated for OIC or IBS-C. Of the patients treated for CIC, women were more likely to report nausea than men, which was not the case for patients treated for OIC or IBS-C.
The majority of patients who experienced nausea reported only a single incident; most patients reported mild-to-moderate nausea that allowed them complete the treatment course. Nausea commonly occurred early in therapy, within a median of 2 to 3 days for CIC or IBS-C. Based on the findings of this study and currently recommended dosing strategies, patients should take lubiprostone with food and water to reduce the incidence of nausea. If nausea does occur, reducing the dose or temporarily discontinuing therapy may help alleviate the symptoms. It may be advisable to offer additional counseling regarding proper administration of lubiprostone to patients who were at higher risk of developing nausea in this study (i.e., women with CIC, patients younger than 65 years of age who have CIC, and patients with a body mass index less than 25 kg/m2 with IBS-C).25,27
Additional adverse effects of lubiprostone include hypotension, syncope, or transient dyspnea that can occur with the first dose of lubiprostone.25 These symptoms generally resolve quickly, within 3 hours for dyspnea, and prior to the next dose for hypotension and syncope. If these symptoms recur, patients should be advised to discontinue therapy, and switching to another treatment is warranted.
Linaclotide
Linaclotide is a poorly absorbed amino acid peptide that acts as a guanylate cyclase-C agonist at the intestinal lumen, increases cyclic GMP (cGMP) concentrations, activates the cystic fibrosis transmembrane conductance regulator (CFTR), thereby increasing chloride and fluid secretion into the GI lumen.13 The accumulation of extracellular cGMP is thought to reduce visceral afferent nerve activity and hypersensitivity and pain, hallmark symptoms of IBS. Linaclotide is approved by FDA for treatment of CIC as well as IBS-C in adults.28
Linaclotide was evaluated for the treatment of CIC in two trials that randomized 1,276 patients to receive linaclotide 145 mcg, linaclotide 290 mcg, or placebo once daily.29 The primary outcome was achievement of at least 3 complete spontaneous bowel movements (CSBMs, a bowel movement that occurs without use of any laxative, enema, or suppository and is accompanied by a feeling of complete evacuation) per week and an increase of at least 1 CSBM per week from baseline for at least 9 of the 12 weeks of the study period. The primary endpoint was achieved in the two clinical trials by 16% and 21% of patients receiving linaclotide 145 mcg, 19.4% and 21.3% of patients receiving linaclotide 290 mcg, and 3.3% and 6% of patients receiving placebo, respectively (P <0.01 for all comparisons versus placebo). In addition, both doses of linaclotide significantly improved stool consistency, straining severity, abdominal discomfort, bloating, and constipation severity compared with placebo at 12 weeks (P < 0.05 for all comparisons versus placebo).
Adverse effects were reported by 52% of patients in the placebo group, and 60.5% and 55.7% of patients in the linaclotide 145 mcg and 290 mcg groups, respectively. The most common adverse effect was diarrhea, which resulted in treatment discontinuation in 4.7% and 3.8% of patients receiving linaclotide 145 mcg and 290 mcg, respectively, compared with 0.5% of patients receiving placebo.29
Linaclotide was also assessed for the treatment of IBS-C in two randomized, placebo-controlled trials with a total of 1,604 patients.30,31 Both trials evaluated whether participants were IBS-C responders at 12 weeks, defined by FDA as those with improvement in average daily worst abdominal pain score of at least 30% from baseline, and an increase if at least 1 CSBM from baseline in the same week for at least 6 of the 12 weeks assessed. Additional improvements in abdominal pain and CSBMs for 9 of the 12 weeks were also assessed.
In the two trials, 33.6% and 33.7% of patients receiving linaclotide 290 mcg daily achieved the primary endpoint in the two trials, compared with 31% and 13.9% of patients receiving placebo (P <0.0001). Additional endpoints evaluating pain, frequency, and stool consistency and bowel symptoms were all significantly improved with linaclotide compared with placebo over 12 and 26 weeks in the two trials.30,31
Diarrhea was the most commonly reported adverse effect and resulted in treatment discontinuation in 5.7% and 4.5% of patients receiving linaclotide compared with 0.3% and 0.2% of patients receiving placebo. All other adverse effects were reported equally between treatment and placebo groups.30,31
Plecanatide
Plecanatide is a structural analog of uroguanylin, an endogenous guanylate cyclase-C agonist. Similar to linaclotide, plecanatide increases cGMP production, which stimulates the CFTR and increases chloride and fluid secretion into the GI lumen.32
Plecanatide is approved by FDA for the treatment of CIC based on two randomized, placebo- controlled 12-week trials of plecanatide 3 mg or 6 mg daily.33,34 The primary outcome in both trials was durable overall CSBM response, defined as the achievement of weekly response (at least 3 CSBMs within a week, and an increase of at least 1 CSBM from baseline in the same week) for at least 9 of the 12 weeks and in at least 3 of the final 4 weeks of the trial.
The first trial randomized 1,337 patients and found that 20.1%, 20%, and 12.8% of patients achieved the primary outcome with plecanatide 3 mg, plecanatide 6 mg, and placebo, respectively (P = 0.004 for both plecanatide groups compared with placebo).33 The second trial had similar results for 1,394 patients, with 21%, 19.5%, and 10.2% of patients achieving the primary endpoint, respectively (P <0.001 for both plecanatide groups compared with placebo).34
The most common adverse effect in the trials was diarrhea, which occurred in 3.2% and 5.9% of patients receiving plecanatide 3 mg, 4.5% and 5.7% of patients receiving 6 mg, and 1.3% of patients receiving placebo in both trials.33,34 Diarrhea is listed as a warning in the product labeling, which recommends immediate discontinuation of plecanatide and rehydration of the patient if severe diarrhea occurs.32
Plecanatide received FDA approval for the treatment of IBS-C in January 2018.32,35 This approval was based on findings from two randomized, placebo-controlled 12-week trials of plecanatide 3 mg or 6 mg in approximately 2,100 patients with IBS-C. The primary endpoint was the percentage of patients who were overall responders as defined by the FDA (at least a 30% reduction in worst abdominal pain and an increase of at least 1 CSBM from baseline in the same week for at least 6 of the 12 treatment weeks). Significantly more patients receiving plecanatide were overall responders compared with placebo (30.2% and 21.5% of patients receiving plecanatide and 17.8% and 14.2% of patients receiving placebo achieving overall response in the two studies, respectively; P <0.05 compared with placebo).32,35
Diarrhea was the most commonly reported adverse effect (4.3% plecanatide, 1% placebo) which led to discontinuation in 1.2% of patients receiving plecanatide compared with no patients receiving placebo. Overall discontinuation rates were low in the trials (2.5% plecanatide and 0.4% placebo). Although all of the phase 3 trials evaluated plecanatide 3 mg and 6 mg doses, the approved dose is 3 mg once daily for both CIC and IBS-C.32,35
Head-to-head trials of treatments for CIC or IBS-C would allow clinicians and patients to make informed decisions regarding therapy choices; however, these are lacking. In lieu of studies that directly compare treatments for CIC, a network meta-analysis was conducted to ascertain which treatment approaches are most effective for patients with CIC.36 Network meta-analyses, similar to direct meta-analyses, provide high-quality evidence when study heterogeneity and effect modifiers are adequately controlled.37,38 Network meta-analyses are statistical models that allow for indirect comparisons between treatments in different trials if at least one of the treatments is common to two or more trials. This allows treatments in separate trials to be compared in situations where head-to-head trials have not been conducted.
Nelson and colleagues found, through direct meta-analysis, that in 21 randomized controlled trials enrolling 9,189 patients, bisacodyl, sodium picosulfate, prucalopride, and velusetrag achieved the primary endpoint of 3 or more CSBMs per week compared with placebo. Through network meta-analysis, none of the agents evaluated (prucalopride, lubiprostone, linaclotide, tegaserod, velusetrag, elobixibat, bisacodyl, or sodium picosulfate) was shown to be superior with regard to CSBM per week. Bisacodyl performed better than the other agents for the secondary endpoint of change in baseline number of SBMs per week.
Given these results, the choice of therapy for a patient with CIC or IBS-C should be made by the healthcare provider and the patient based on the patient's treatment goals, and medication- specific attributes such as dosing regimens, adverse effect profiles, cost of therapy, accessibility (OTC versus prescription), and previous patient and healthcare provider experience with available treatment options.
Emerging Therapies
Serotonin 5-HT4 receptors have been a target for drug therapy of CIC and IBS-C in the past. Activation of 5-HT4 receptors in the GI tract results in increased mucosal secretion and gut motility.6,24,40 Two agents in this class — cisapride in 2000 and tegaserod in 2007 — were withdrawn from the U.S. market because of adverse cardiovascular effects, including fatal arrhythmias, heart attacks, and strokes. These effects resulted from the drugs' affinity for hERG-K+ cardiac channels.
The 5-HT4 agonists currently in development are highly selective with low affinity for hERG-K+ cardiac receptors, limiting their potential for cardiotoxicity. Agents in development include prucalopride, velusetrag, and naronapride. Prucalopride is currently available for treatment of women with CIC in Canada and Europe.41
Bile acids have garnered interest in the area of CIC and IBS-C. Elboxibat is a bile acid transporter inhibitor that produces increased concentrations of bile acids within the GI lumen.41,42 These promote colonic secretions and contractions and accelerate transit. Elboxibat is currently being evaluated in the treatment of CIC.
Tenapanor is an inhibitor of the sodium-hydrogen exchanger 3 (NHE-3) channel, which plays a role in the regulation of intestinal sodium and water uptake throughout the GI tract. This results in increased fluid volume and faster intestinal transit. In preliminary trials, tenapanor has been minimally absorbed and well tolerated, with diarrhea being its most common adverse effect.6,24
THE ROLE OF THE PHARMACIST IN MANAGING CONSTIPATION
Constipation related to CIC and IBS-C is a common complaint. Pharmacists are in a unique position as the most accessible healthcare provider for patients seeking advice regarding self- care or prescription therapies for constipation. Unfortunately, even though many patients would like to receive education and advice about this condition from their pharmacists, they frequently do not actively seek it.8 This may be due to embarrassment on the patient's part, or lack of awareness of the help and support the pharmacist can provide.
Patients with IBS-C report that having a healthcare provider listen, provide empathy, and provide education regarding their disease and therapy options would maximize their relationship with the healthcare provider.10 In addition, patients' IBS symptoms may improve further with reassurance from a healthcare provider.41,43
Pharmacists and other health professionals can use structured patient counseling approaches and objective tools such as the BSFS to educate patients about their constipation and its therapies and thereby overcome barriers to treatment. Box 2 provides a list of online resources geared toward patients with CIC or IBS that may aid the pharmacist when educating patients about their symptoms.
Box 2. Patient Educational Resources Regarding CIC and IBS
American Gastroenterological Association (AGA) Patient Education Resources provide definitions and information about various gastrointestinal diseases and their treatments and patient summaries of treatment guidelines.
- AGA Patient Education Resources for IBS http://www.gastro.org/patient-care/conditions- diseases/irritable-bowel-syndrome
- AGA Patient Education Resources for Constipation http://www.gastro.org/patient- care/conditions-diseases/constipation
U.S. National Library of Medicine Medline Plus includes educational material regarding a variety of diseases as well as links to other reputable resources for patient education.
- Medline Plus entry for Constipation https://medlineplus.gov/constipation.html Medline Plus entry for IBS https://medlineplus.gov/irritablebowelsyndrome.html
American College of Gastroenterology (ACG) Patient Resource Center provides patient education resources including links to other resources and some videos and podcasts geared toward patients.
- ACG Patient Resources for Constipation http://patients.gi.org/topics/constipation- and-defection-problems/#tabs2
- ACG Patient Resources for IBS http://patients.gi.org/topics/irritable-bowel- syndrome/#tabs1
|
Figure 2 provides a proposed treatment algorithm with structured questions a pharmacist may ask a patient to rule out alarm symptoms and begin an open conversation regarding the patient's constipation. The algorithm also provides a proposed stepwise approach to the management of CIC and IBS-C. Each step in the algorithm should be continued for several (8 to 12) weeks to ensure an adequate treatment trial prior to considering a patient as having failed a given therapy. Intolerable adverse effects would constitute therapy failure provided treatment was administered as recommended (e.g., slow titration of fiber to avoid GI adverse effects, administration of lubiprostone with food to reduce nausea symptoms).
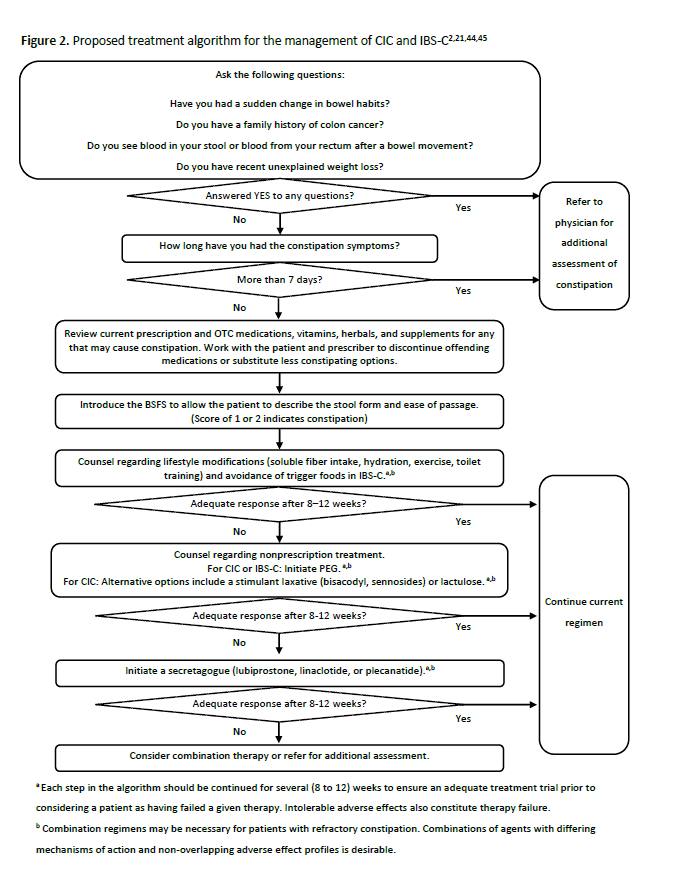
Patients who fail to respond adequately to a given therapy may benefit from combination therapy. Agents with differing mechanisms of action and nonoverlapping adverse effect profiles can be used together for increased effects. In addition to therapies directly targeting the GI tract, patients with IBS-C may benefit from adjunctive therapies such as tricyclic antidepressants, selective serotonin reuptake inhibitors, antispasmodic agents, probiotics or peppermint oil, as well as psychotherapy. Severe refractory CIC may require surgical intervention.
Treatment of CIC and IBS-C is often an ongoing trial-and-adjustment process. No single regimen or combination therapy works for all patients. Treatment should be tailored to the response and adverse effects experienced by each individual.
CONCLUSION
The pharmacist plays an important role for patients with CIC or IBS-C, including provision of information about constipation, help in choosing nonprescription treatments, and education regarding prescription therapies. Pharmacists can provide education and encouragement regarding lifestyle changes. Pharmacists can also provide empathy and support through structured patient counseling and use of tools such as the BSFS.
REFERENCES
- Suares NC, Ford AC. Prevalence of, and risk factors for, chronic idiopathic constipation in the community: systematic review and meta-analysis. Am J Gastroenterol. 2011;106(9):1582-1591.
- Lindberg G, Hamid SS, Malfertheiner P, et al. World Gastroenterology Organisation global guideline: constipation—a global perspective. J Clin Gastroenterol. 2011;45(6):483-487.
- Mearin F, Lacy BE, Chang L, et al. Bowel disorders. Gastroenterology. 2016;150:1392-1407.
- Soh AYS, Kang JY, Siah KTH, Scarpignato C, Gwee KA. Searching for a definition for pharmacologically refractory constipation: a systematic review. J Gastroenterol Hepatol. 2017. [epub ahead of print].
- Defrees DN, Bailey J. Irritable bowel syndrome: epidemiology, pathophysiology, diagnosis, and treatment. Prim Care. 2017;44(4):655-671.
- Hayat U, Dugum M, Garg S. Chronic constipation: update on management. Cleve Clin J Med. 2017;84(5):397-408.
- Schmulson MJ, Drossman DA. What is new in Rome IV. J Neurogastroenterol Motil. 2017;23(2):151-163.
- Shibata K, Matsumoto A, Nakagawa A, et al. Use of pharmacist consultations for nonprescription laxatives in Japan: an online survey. Biol Pharm Bull. 2016;39(11):1767-1773.
- Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32(9):920-924.
- Halpert A, Godena E. Irritable bowel syndrome patients' perspectives on their relationships with healthcare providers. Scand J Gastroenterol. 2011;46(7-8):823-830.
- Salvo MC, Cannon-Breland ML. Motivational interviewing for medication adherence. J Am Pharm Assoc. 2015;55(4):e354-3e61.
- Rutter PM, Horsley E, Brown DT. Evaluation of community pharmacists' recommendations to standardized patient scenarios. Ann Pharmacother. 2004;38(6):1080-1085.
- Rao SS, Rattanakovit K, Patcharatrakul T. Diagnosis and management of chronic constipation in adults. Nat Rev Gastroenterol Hepatol. 2016;13(5):295-305.
- Bharucha AE, Dorn SD, Lembo A, Pressman A. American Gastroenterological Association medical position statement on constipation. Gastroenterology. 2013;144(1):211-217.
- Johanson JF, Kralstein J. Chronic constipation: a survey of the patient perspective. Aliment Pharmacol Ther. 2007;25(5):599-608.
- Muller-Lissner SA, Kamm MA, Scarpignato C, Wald A. Myths and misconceptions about chronic constipation. Am J Gastroenterol. 2005;100(1):232-242.
- Schuster BG, Kosar L, Kamrul R. Constipation in older adults: stepwise approach to keep things moving. Can Fam Physician. 2015;61(2):152-158.
- Lee HJ, Choi JK, Ryu HS, et al. Therapeutic modulation of gut microbiota in functional bowel disorders. J Neurogastroenterol Motil. 2017;23(1):9-19.
- Harris LA, Baffy N. Modulation of the gut microbiota: a focus on treatments for irritable bowel syndrome. Postgrad Med. 2017;129(8):872-888.
- Staudacher HM, Kurien M, Whelan K. Nutritional implications of dietary interventions for managing gastrointestinal disorders. Curr Opin Gastroenterol. 2018;34(2):105-111.
- Ford AC, Moayyedi P, Lacy BE, et al. American College of Gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am J Gastroenterol. 2014;109(Suppl 1):S2-S26.
- Moayyedi P, Quigley EM, Lacy BE, et al. The effect of fiber supplementation on irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol. 2014;109(9):1367-1374.
- Schoenfeld PS. Advances in IBS 2016: a review of current and emerging data. Gastroenterol Hepatol. 2016;12(8 Suppl 3):1-11.
- Thomas RH, Luthin DR. Current and emerging treatments for irritable bowel syndrome with constipation and chronic idiopathic constipation: focus on prosecretory agents. Pharmacotherapy. 2015;35(6):613-630.
- Amitiza® [package insert]. Bethesda, MD: Sucampo Pharmaceuticals, Inc.; 2017.
- Fukudo S, Hongo M, Kaneko H, Takano M, Ueno R. Lubiprostone increases spontaneous bowel movement frequency and quality of life in patients with chronic idiopathic constipation. Clin Gastroenterol Hepatol. 2015;13(2):294-301.e295.
- Cryer B, Drossman DA, Chey WD, Webster L, Habibi S, Wang M. Analysis of nausea in clinical studies of lubiprostone for the treatment of constipation disorders. Dig Dis Sci. 2017;62(12):3568-3578.
- Linzess® [package insert]. Cambridge, MA: Ironwood Pharmaceuticals, Inc.; 2017.
- Lembo AJ, Schneier HA, Shiff SJ, et al. Two randomized trials of linaclotide for chronic constipation. N Engl J Med. 2011;365(6):527-536.
- Rao S, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol. 2012;107(11):1714-1724.
- Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol. 2012;107(11):1702-1712.
- Trulance® [package insert]. New York, NY: Synergy Pharmaceuticals, Inc.; 2018.
- DeMicco M, Barrow L, Hickey B, Shailubhai K, Griffin P. Randomized clinical trial: efficacy and safety of plecanatide in the treatment of chronic idiopathic constipation. Ther Adv Gastroenterol. 2017;10(11):837-851.
- Miner PB, Koltun WD, Wiener GJ, et al. A randomized phase III clinical trial of plecanatide, a uroguanylin analog, in patients with chronic idiopathic constipation. Am J Gastroenterol. 2017;112(4):613-621.
- Synergy Pharmaceuticals Announces FDA Approval of Trulance® (Plecanatide) for the Treatment of Irritable Bowel Syndrome With Constipation (IBS-C) in adults. January 25, 2018. Available at: https://ir.synergypharma.com/press-releases/detail/1861/synergy-pharmaceuticals-announces-fda-approval-of. Accessed January 27, 2018.
- Nelson AD, Camilleri M, Chirapongsathorn S, et al. Comparison of efficacy of pharmacological treatments for chronic idiopathic constipation: a systematic review and network meta-analysis. Gut. 2017;66(9):1611-1622.
- Leucht S, Chaimani A, Cipriani AS, Davis JM, Furukawa TA, Salanti G. Network meta-analyses should be the highest level of evidence in treatment guidelines. Eur Arch Psychiatry Clin Neurosci. 2016;266(6):477-480.
- Salanti G, Del Giovane C, Chaimani A, Caldwell DM, Higgins JP. Evaluating the quality of evidence from a network meta-analysis. PloS One. 2014;9(7):e99682.
- Chey WD, Lembo AJ, Rosenbaum DP. Tenapanor treatment of patients with constipation-predominant irritable bowel syndrome: a phase 2, randomized, placebo-controlled efficacy and safety trial. Am J Gastroenterol. 2017;112(5):763-774.
- Bharucha AE, Wouters MM, Tack J. Existing and emerging therapies for managing constipation and diarrhea. Curr Opin Pharmacol. 2017;37:158-166.
- Ford AC, Lacy BE, Talley NJ. Irritable bowel syndrome. N Engl J Med. 2017;376(26):2566-2578.
- Mosinska P, Salaga M, Fichna J. Novel investigational drugs for constipation-predominant irritable bowel syndrome: a review. Expert Opin Investig Drugs. 2016;25(3):275-286.
- Kaptchuk TJ, Friedlander E, Kelley JM, et al. Placebos without deception: a randomized controlled trial in irritable bowel syndrome. PloS One. 2010;5(12):e15591.
- Tse Y, Armstrong D, Andrews CN, et al. Treatment algorithm for chronic idiopathic constipation and constipation-predominant irritable bowel syndrome derived from a Canadian national survey and needs assessment on choices of therapeutic agents. Can J Gastroenterol Hepatol. 2017;2017:8612189.
- Wald A. Constipation: advances in diagnosis and treatment. JAMA. 2016;315(2):185-191.
Back to Top