
Expired activity
Please go to the PowerPak
homepage and select a course.
Module 3. Overview of Non-Insulin Injectable Glucose-Lowering Agents
INTRODUCTION
Diabetes is an ever-growing health care concern in the United States (U.S.), associated with significant morbidity and high health care costs. In 2015, an estimated 30.3 million adults (9.4 percent of the U.S. population) in the U.S. had diabetes.1 In 2012, the total cost associated with diabetes was $245 billion.1 It is essential that health care professionals understand how to appropriately manage the disease.
Guidelines for the management of patients with diabetes are routinely released and updated. The goal of this course is to help pharmacists understand therapies used to treat diabetes and remain current with practice standards of diabetes care. To do so, this module relies on the full guidelines issued in 2019 by the American Diabetes Association (ADA) as well as a 2019 consensus statement by the American Association of Clinical Endocrinologists and the American College of Endocrinology (AACE/ACE).2,3
The focus of this module is on the non-insulin injectable drugs currently available in the U.S., a topic that encompasses two classes of medication: glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and amylin receptor agonists. Additionally, two products on the market contain a fixed-dose combination of a long-acting insulin and a GLP-1 RA. These combination products will be discussed herein, with a more detailed discussion of insulin therapy provided in another module.
GENERAL CONSIDERATIONS FOR INJECTABLE THERAPIES
All of the products discussed in this module are administered by subcutaneous injection. As such, it is important to consider key barriers patients face when prescribed injectable medications. Fear of needles may make some patients hesitant to initiate these therapies, but patient-centered counseling can help overcome this fear. Pharmacists should also carefully assess patients' abilities to administer the injection. Do they understand the appropriate administration technique? Are they capable of successfully reconstituting a medication that does not come premixed? Does the patient have enough dexterity to use a syringe or prefilled pen device? Detailed recommendations for addressing patient barriers are available in the insulin therapy module. Tips for administration of each product in this module are shown in Table 1.
| Table 1. Administration Tips for GLP-1 RAs.4-11 |
| Generic |
Brand |
Devices Available |
Administration Instructions |
| Exenatide (regular release) |
Byetta |
Pen-injector
(5 mcg or 10 mcg) |
Requires pen needles; Dial dose knob until horizontal arrow appears; Pull dose knob out until it stops and vertical arrow appears; Dial dose knob until dose appears; Insert needle into subcutaneous tissue and push dose knob, hold for 5 seconds; Reset device by dialing dose knob until horizontal arrow appears |
| Exenatide (extended release) |
Bydureon |
Vial kit
(2 mg) |
Break off syringe cap and remove vial lid; Connect vial and syringe via orange connector and inject diluent into vial; Shake connected vial and syringe until medication is mixed well; Draw reconstituted medication into syringe by pulling plunger beyond the dashed dose line; Unscrew the orange connector to remove vial from syringe and attach the provided needle to syringe; Push syringe plunger to the dashed dose line; Insert needle into subcutaneous tissue and inject dose |
Pen-injector
(2 mg) |
Leave pen at room temperature for 15 minutes before use; Screw provided needle onto pen; Turn white knob until green label disappears and device clicks; Tap pen firmly against the palm up to 80 times, rotating every 10 taps to ensure medication is well mixed; Turn the white knob until orange label disappears and injection button is released; Insert needle into subcutaneous tissue and press injection button until it clicks, hold for 10 seconds |
| Bydureon BCise |
Auto-injector
(2 mg) |
Leave autoinjector at room temperature for 15 minutes before use; Shake autoinjector vigorously in up-and-down motion for at least 15 seconds to ensure medication is well mixed; Turn knob from lock to unlock until it clicks; Unscrew orange cap, allowing green needle shield to pop up; Push autoinjector against skin, device will click when injection begins, hold for 15 seconds |
| Liraglutide |
Victoza |
Pen-injector
(18 mg/3 mL) |
Requires pen needles; Turn dose selector to prescribed dose (0.6, 1.2, or 1.8 mg); Insert needle into subcutaneous tissue, press and hold dose button until pointer aligns with 0 mg, continue to hold for an additional 6 seconds |
| Dulaglutide |
Trulicity |
Pen-injector
(0.75 mg/0.5 mL or 1.5 mg/0.5 mL) |
With pen locked, pull off base cap; Place clear base flat against injection site; Unlock pen by turning the lock ring; Press and hold green injection button, initial click begins injection; Hold pen firmly against skin until second click (approximately 5-10 seconds) |
| Semaglutide |
Ozempic |
Pen-injector
(1 mg per dose or 0.25 mg/0.5 mg per dose) |
See Victoza instructions above; Needles are provided with Ozempic; Ozempic doses are dialed in mg of semaglutide (0.25, 0.5, or 1 mg) |
Insulin degludec/
liraglutide |
Xultophy |
Pen-injector
(100 units/3.6 mg per mL) |
See Victoza instructions above; Xultophy doses are dialed in units of basal insulin |
Insulin glargine/
lixisenatide |
Soliqua |
Pen-injector
(100 units/33 mcg per mL) |
Requires pen needles; Dial dose selector to prescribed dose of basal insulin; Insert needle into subcutaneous tissue, press and hold dose button until pointer aligns with 0 mg, continue to hold for an additional 10 seconds |
| Note: Priming instructions vary by device and are not included in this table. Counsel patients to review full instructions provided by manufacturer to ensure proper priming and administration. |
For non-insulin injectables, cost may be an additional barrier to patient acceptance and compliance. No generic products are presently available in this category. Formulary restrictions may dictate which medications in this class are available to a particular patient, and discount, rebate, or patient-assistance programs may be necessary to increase affordability.
Finally, pharmacists and pharmacy technicians should be aware that patients with diabetes are increasingly prescribed multiple injectable therapies, including both insulin and non-insulin medications. The variety of brand names, dosing regimens, and administration devices can be confusing for clinicians and patients alike. Ensuring that patients understand the purpose of each medication and its place in therapy is crucial to preventing administration errors, reducing the risk of hypoglycemia, and optimizing glycemic control.
NORMAL GLUCOSE HOMEOSTASIS
To understand the mechanisms of action of the agents discussed in this module, it is necessary to review the normal physiology surrounding blood glucose regulation. More information is available in the pathophysiology module. Glucose in the body comes from two sources: endogenous glucose production in the liver and glucose absorbed through dietary intake. Levels of glucose in the blood are kept in check primarily by the opposing hormones insulin and glucagon. In general, insulin acts to suppress hepatic glucose production and promote glucose uptake by peripheral tissue (primarily skeletal muscle and adipose tissue). Conversely, glucagon acts to increase hepatic glucose production and reduce glucose uptake by peripheral tissues.12
When a person ingests food, the neuroendocrine cells of the gastrointestinal tract secrete hormones called incretins. When blood glucose levels rise above the fasting level, these incretins bind to receptors on α and β cells of the pancreas, promoting secretion of insulin from the β cells and suppressing secretion of glucagon from the α cells. The result is a net increase in glucose uptake by peripheral tissue and decrease in glucose production by the liver. Incretins also slow gastric emptying, which delays the absorption of glucose.12 Endogenous incretins have very short half-lives and are degraded by the enzyme dipeptidyl peptidase 4 (DPP-4). This short half-life makes the naturally occurring incretins unsuitable for administration as diabetes management medications. However, synthetic incretins with much longer half-lives and molecules capable of activating incretin receptors have been developed to promote the incretin effect in patients with diabetes. Though not discussed in this module, oral inhibitors of the enzyme DPP-4 have also been developed to help manage diabetes.13
GLUCAGON-LIKE PEPTIDE-1 RECEPTOR AGONISTS (GLP-1 RAS)
Glucagon-like peptide-1 (GLP-1) is the most potent incretin secreted by the gastrointestinal tract in response to food ingestion.12 In addition to stimulating insulin secretion and suppressing glucagon secretion, GLP-1 acts to slow gastric emptying and reduce appetite, which gives GLP-1 the added benefit of promoting weight loss.14 The actions of GLP-1 are mediated by interaction with an incretin receptor called the GLP-1 receptor. As a result, this receptor has been the target of several agents used to manage diabetes.
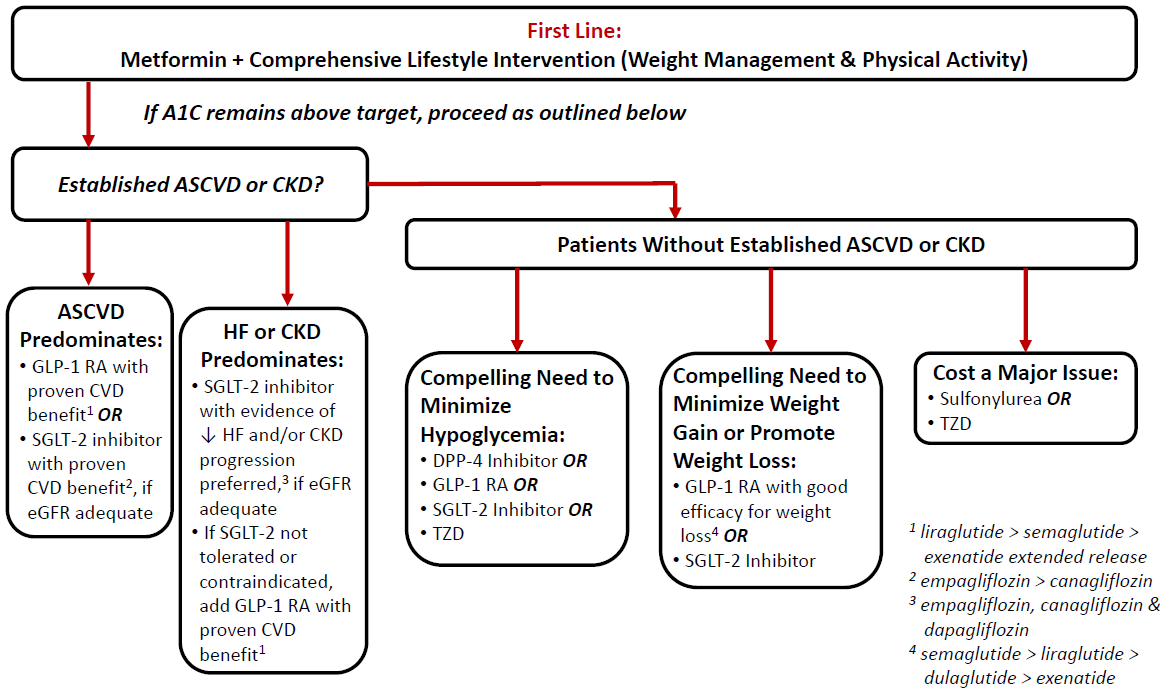
GLP-1 RAs are approved by the U.S. Food and Drug Administration (FDA) for use in the treatment of type 2 diabetes (T2D) in adults. Although studies are ongoing in other patient populations, the available products are not yet approved for patients less than 18 years old or patients with type 1 diabetes. The ADA guidelines (see Figure 1) recommend the GLP-1 RAs as an option as add-on to metformin based on several considerations. 2 First, GLP-1 RAs with proven cardiovascular benefit (currently ranked as liraglutide > semaglutide > exenatide extended release) are recommended as options for add-on to metformin in patients with established atherosclerotic cardiovascular disease (ASCVD). They are additionally recommended in patients in whom heart failure (HF) or chronic kidney disease (CKD) predominates if the patient cannot take an SGLT2 inhibitor due to impaired renal function. For patients who do not have established ASCVD or CKD, GLP-1 RAs are additionally recommended by the ADA for consideration as add-on to metformin when there is a compelling need to minimize hypoglycemia risk or promote weight loss. Notably, the 2019 ADA Standards of Care additionally state that GLP-1 RAs are preferred to insulin in most patients who need the greater glucose-lowering effect of an injectable medication.2 The AACE/ACE Comprehensive Type 2 Diabetes Management Algorithm similarly gives GLP-1 RAs high level recommendations in the management of T2D.3 GLP-1 RAs are noted within the guideline as a second-line monotherapy (following metformin) as well as a first-line option for therapy escalation beyond monotherapy in a patient not already receiving a GLP-1 RA.3 Similar to ADA, the AACE/ACE algorithm also considers comorbidities such as cardiovascular disease (CVD) and CKD. At the point of monotherapy, AACE/ACE states that GLP-1 RAs or SGLT2 inhibitors with proven CVD and CKD benefits are preferred as first-line monotherapy in patients with those complications.3 As with all diabetes medications, GLP-1 RAs are always considered adjuncts to lifestyle modification.2,3 Due to the mutual impact of GLP-1 RAs and DPP-4 inhibitors on the incretin system, the two classes of medications are not recommended for combined use.2,3
|
Figure 1.2
|
The most common side effects associated with GLP-1 RAs are nausea, diarrhea, and headache, commonly experienced early in therapy but subsiding over time.15 These side effects can usually be reduced by initiating the medication at a low dose and titrating slowly. In addition, nausea may be reduced by eating smaller portions and reducing fat intake. The different agents vary slightly in the intensity of their side effect profile, but patients should generally expect some gastrointestinal side effects at the initiation of therapy.
All GLP-1 RAs slow gastric emptying to some degree. The shorter-acting agents, however, affect gastric emptying to a greater degree. As such, there is a general precaution against their use in patients with gastrointestinal disorders, including gastroparesis.16-22 The effect on gastric emptying also has the potential to reduce the absorption of orally administered medications. Although the manufacturers do not report any clinically significant interactions as a result of this effect, caution is advised when co-administering GLP-1 RAs with oral medications that have a narrow therapeutic index or require close clinical monitoring.16-22
The risk of hypoglycemia with GLP-1 RAs is low when used as monotherapy.2 However, the risk of hypoglycemia increases when GLP-1 RAs are administered concomitantly with insulin or insulin secretagogues. The manufacturers of GLP1-RAs typically recommend lowering doses of insulin and insulin secretagogues at the initiation of GLP-1 RAs to reduce the risk of hypoglycemia.16-22
Among rarer, more serious side effects are reports that GLP-1 RAs as a class may be associated with the development of pancreatitis. Causality has not been established, but these agents should be used cautiously in patients with a history of pancreatitis. Studies involving rats have also shown a relationship between GLP-1 RAs and the development of thyroid c-cell tumors, including medullary thyroid carcinoma (MTC). As such, these agents are generally contraindicated in patients with a personal or family history of MTC or in patients with multiple endocrine neoplasia syndrome type 2.6-22
There are five GLP-1 RA products currently on the market: exenatide, exenatide extended release, liraglutide, dulaglutide, and semaglutide. A sixth agent, lixisenatide, is FDA-approved but only currently available in a fixed-dose combination with insulin glargine. Another FDA-approved GLP-1 RA, albiglutide (Tanzeum), was discontinued by the manufacturer in 2017 due to lagging sales and increased competition.
Exenatide
Exenatide is a synthetic version of a peptide known as exendin-4, which was originally discovered in the saliva of the Gila monster. Exenatide is capable of activating GLP-1 receptors, but its modified amino acid structure allows it to resist degradation by DPP-4 for much longer than endogenous GLP-1.13
Exenatide is available in 2 formulations: regular (Byetta) and extended-release (Bydureon). The regular-release formulation is available as a fixed-dose prefilled pen device in two doses: 5 mcg and 10 mcg. Each pen contains 60 doses. The initiation dose is 5 mcg twice daily, to be injected 30 to 60 minutes prior to a meal. After 1 month of therapy the dose may be increased to 10 mcg twice daily as necessary and as tolerated.16
Two extended-release formulations of exenatide are currently marketed, differing primarily in administration device. The original formulation, Bydureon, is available both as a single-dose, 2 mg vial requiring reconstitution with the supplied diluent and as a single-dose, prefilled pen-injector. A new formulation, Bydureon BCise, is available as an auto-injector. See Table 1 for the administration differences in these products. Regardless of the extended release exenatide formulation prescribed, the dose is 2 mg injected once weekly with or without food. A missed weekly dose should be administered as soon as it is remembered within the next 3 days. If it has been more than 3 days since the missed dose, the patient should skip the missed dose and resume the regular schedule.17,18
It is important to consider the effect of each exenatide formulation on blood glucose, A1C, and weight. The regular-release formulation has a stronger effect on postprandial blood glucose levels than the extended-release formulation. Conversely, the extended-release formulation has a stronger effect on fasting blood glucose levels than the regular-release formulation. Exenatide is not labeled for use as a weight-loss aid; however, weight loss with exenatide was noted in the clinical trials of all three formulations. The mean weight loss with exenatide monotherapy at 24 to 30 weeks ranged from about 3 to 6 lb in clinical trials.16-18
The manufacturer recommends that oral contraceptives be taken at least 1 hour prior to injection of twice daily exenatide.16 Postmarketing studies have shown that exenatide may increase the risk of bleeding with warfarin. The manufacturer recommends increased monitoring of INR upon initiation.16-18
The most common adverse effects associated with exenatide use include nausea, diarrhea, headache, vomiting, constipation, and dyspepsia. These gastrointestinal side effects usually subside over time.16-18 The regular release formulation has also been associated with feeling jittery, dizziness, and asthenia and carries a higher risk of hypoglycemia than the extended-release formulation.16 The extended-release formulation is more likely to cause injection site reactions (pruritus with or without nodules).17,18 Exenatide should be used cautiously in patients with creatinine clearance (CrCl) of 30 to 50 mL/min and should not be used in patients with CrCl less than 30 mL/min.16-18 Exenatide extended-release has been evaluated for long-term safety in a 5-year, uncontrolled study. Patients taking the study medication long-term showed sustained reductions in A1C without any major cases of hypoglycemia or alarming safety events.23
Liraglutide
Liraglutide is a synthetic analog of human GLP-1. The two molecules are almost identical, but liraglutide contains a hydrophobic group that allows it to bind to albumin and other plasma proteins, resisting degradation by DPP-4 and substantially increasing its half-life.13 Liraglutide is marketed under two brand names. The original product, Victoza, is approved for glycemic control in adults with T2D and to reduce the risk of major adverse cardiovascular events in adults with T2D and established cardiovascular disease.19 Liraglutide is also available as Saxenda, an injectable weight-loss medication. The dose of Saxenda is higher than the dose of Victoza.24 Pharmacists should remain alert to the differing products and indications to ensure the correct dose of liraglutide is provided to each patient.
Liraglutide for glycemic control is available as 6 mg/mL solution in a prefilled pen device, which is capable of delivering doses of 0.6 mg, 1.2 mg, and 1.8 mg. Liraglutide should be initiated at a dose of 0.6 mg injected once daily with or without food. This low dose is not considered therapeutic for glycemic control, though some patients may see an effect. Initiation at this low dose helps to reduce the incidence of gastrointestinal adverse effects. After 1 week of therapy, the dose should be increased to 1.2 mg injected once daily. This dose is considered therapeutic but may be increased to 1.8 mg injected once daily if the patient has not met glycemic targets. It is important to note that if a patient misses more than 3 days of consecutive dosing, therapy should be re-initiated at the 0.6 mg daily dose. Failure to do so may result in unacceptable gastrointestinal adverse effects.19
In a clinical trial of patients with diabetes using liraglutide monotherapy for glycemic control, the mean weight loss at 52 weeks was about 6 lb.19
Liraglutide is unique among GLP-1 RAs in its indication for cardiovascular risk reduction in patients with diabetes and established cardiovascular disease.19 The LEADER trial compared liraglutide 1.8 mg daily to placebo for a median duration of 3.5 years. Liraglutide significantly reduced the time to a first occurrence of non-fatal myocardial infarction, non-fatal stroke, or cardiovascular death, with an estimated hazard ratio of 0.87.19 As a result, the ADA guidelines recommend liraglutide as an option to add to metformin in patients with comorbid diabetes and cardiovascular disease (see Figure 1).2
The most common adverse effects associated with liraglutide use are headache, nausea, diarrhea, and anti-liraglutide antibody formation. The formation of anti-liraglutide antibodies does not appear to reduce the efficacy of liraglutide therapy.19
Dulaglutide
Dulaglutide (Trulicity) is a fusion protein of 2 copies of a human GLP-1 analog fused to human immunoglobulin G4. The GLP-1 proteins have been modified to improve their resistance to degradation by DPP-4.20 These modifications give dulaglutide a very long half-life.
Dulaglutide is available as 0.75 mg/0.5 mL or 1.5 mg/0.5 mL solution in a single-dose prefilled pen. The initiation dose is 0.75 mg injected once weekly with or without food. The dose may be increased to 1.5 mg injected once weekly as tolerated and as necessary for glycemic control. A missed dose should be administered as soon it is remembered within the next 3 days. If it has been more than 3 days since the missed dose, the dose should be skipped and the regular schedule resumed.20
Dulaglutide has been shown to reduce A1C by about 1%.20 Although dulaglutide is not approved for weight loss, trials of dulaglutide monotherapy in patients with diabetes showed a mean weight loss of 3 to 5 lb at 26 weeks.20
The most common adverse effects associated with dulaglutide are nausea, diarrhea, vomiting, abdominal pain, and decreased appetite. In clinical trials, dulaglutide was associated with a mean increase in heart rate of 2–4 beats per minute (bpm). Episodes of sinus tachycardia (increase of 15 bpm or more from baseline) were reported by 2.2% of patients treated with dulaglutide 1.5 mg compared to 0.7% of patients treated with placebo. Additionally, dulaglutide-treated patients experienced a mean increase in PR interval of 2–3 milliseconds, with an increase to at least 220 milliseconds observed in 3.2% of patients treated with dulaglutide 1.5 mg, compared to 0.7% of patients treated with placebo. The clinical implications of these heart rate and PR interval increases are not clear.20
Dulaglutide should be used cautiously in patients with renal impairment because they may experience elevated levels of the medication; however, no dose adjustments are recommended.20
Lixisenatide
Lixisenatide is a peptide that acts as an agonist at the GLP-1 receptor. As noted previously, lixisenatide is approved by the FDA for treatment of T2D but is currently only available in combination with insulin glargine (see the discussion of GLP-1 + basal insulin combinations below). In clinical trials, lixisenatide was dosed once daily within one hour before the first meal of the day. The starting dose is 10 mcg daily, which is increased after 14 days to the maintenance dose of 20 mcg daily.
The most common adverse effects associated with lixisenatide are nausea, vomiting, headache, diarrhea, and dizziness. The majority of the gastrointestinal effects occurred during the first 3 weeks of treatment. Many patients treated with lixisenatide will develop antibodies to the molecule. In clinical trials, the 2.4% of patients with the highest antibody concentrations experienced a reduced glycemic response, and patients who were antibody positive experienced a higher incidence of allergic and injection site reactions. If worsening response or increased adverse effects occur during therapy, consider discontinuing lixisenatide.21
The manufacturer recommends that oral contraceptives be administered at least 1 hour before or 11 hours after each dose of lixisenatide.25 Patients with renal impairment should be monitored closely for adverse reactions and changes in renal function, but no dosage adjustments are recommended. Lixisenatide is not recommended in patients with end-stage renal disease.21
Semaglutide
Semaglutide (Ozempic) is another analog of human GLP-1. Its molecular modifications increase albumin binding, resulting in a decreased renal clearance and protection from degradation by DPP-4. The elimination half-life of semaglutide is extremely long, and the medication remains in circulation for about 5 weeks after the last dose.22
Semaglutide is available in prefilled pens, with one product that delivers 0.25 mg or 0.5 mg per injection and another that delivers only 1 mg per injection. The initial dose is 0.25 mg injected once weekly for 4 weeks. This dose is not effective for glycemic control but reduces the risk of side effects at treatment initiation. After 4 weeks, the dose is increased to 0.5 mg injected once weekly. If additional glycemic control is needed following 4 weeks of the 0.5 mg dose, a maximum dose of 1 mg injected once weekly may be used. Semaglutide should be injected on the same day each week, at any time of day, without regard to meals. Missed doses should be administered as soon as possible within 5 days of the missed dose.22
Although semaglutide is not approved for weight loss, clinical trials of semaglutide monotherapy in patients with T2D resulted in a mean weight loss of around 8 to 10 lb compared to about 2.5 lb for placebo at therapy week 30.22
The most common adverse effects reported in patients treated with semaglutide are nausea, vomiting, diarrhea, abdominal pain, and constipation. A 2-year trial involving patients with T2D and high cardiovascular risk demonstrated an increased risk of diabetic retinopathy among patients treated with semaglutide compared to placebo.26 This finding has not been observed in other clinical trials with semaglutide. The highest risk was to patients with a history of diabetic retinopathy at baseline. The manufacturer recommends that such patients be monitored for progression of diabetic retinopathy when treated with semaglutide.22
GLP-1 RAs should not be used in pregnancy. Due to the long washout period for semaglutide, women of childbearing age should be instructed to discontinue the medication at least 2 months before a planned pregnancy to avoid fetal exposure.22
COMBINATION PRODUCTS: GLP-1 RA WITH BASAL INSULIN
Two products combining a GLP-1 RA and basal insulin in single, fixed-dose injectable are currently approved by the FDA for patients with T2D as adjunct to diet and exercise to improve glycemic control.27,28 Both combination products are dosed based on the insulin component and titrated to effect. As with all insulin-containing products, the most common adverse reaction is hypoglycemia. Patients should be counseled extensively on the detection and treatment of hypoglycemia, as detailed in the insulin therapy module. These products are subject to the risks and benefits of their individual components, which are discussed elsewhere in this course. However, this section will briefly review considerations specific to the combination products.
Insulin Glargine/Lixisenatide
A product combining basal insulin glargine and the GLP-1 RA lixisenatide is marketed under the brand name Soliqua 100/33. The combination contains 100 units of insulin glargine and 33 mcg of lixisenatide per mL and is available in a prefilled pen containing a total of 3 mL.27
If a patient is naïve to a basal insulin and GLP-1 RAs, or is currently receiving less than 30 units of basal insulin, it is recommended that they start the combination product at 15 units (15 units insulin and 5 mcg of lixisenatide) injected once daily. A patient being converted from 30 to 60 units of basal insulin should start the combination product at 30 units injected once daily. The daily dose should be administered within the hour prior to the first meal of the day. Following initiation, the dosage should be titrated by two to four units every week based on blood glucose monitoring results until the desired fasting glucose is achieved. The maximum daily dose is 60 units.27
In addition to hypoglycemia, adverse reactions commonly associated with the combination product include allergic reaction, nausea, nasopharyngitis, diarrhea, upper respiratory tract infection, and headache. Gastrointestinal adverse reactions occur more frequently at the beginning of therapy.27
Insulin Degludec/Liraglutide
A product combining basal insulin degludec and GLP-1 RA liraglutide is marketed under the brand name Xultophy 100/3.6. The combination contains 100 units of insulin degludec and 3.6 mg of liraglutide per mL and is available in a prefilled pen containing a total of 3 mL.28
The recommended starting dose for patients who are naïve to basal insulin and GLP-1 RAs is 10 units (10 units of insulin and 0.36 mg of liraglutide) administered once daily. For patients who are already receiving either basal insulin or a GLP-1 RA, the recommended starting dose is 16 units injected at the same time each day without regard to food. This dose should be titrated by two units every three to four days based on blood glucose monitoring results until the desired fasting glucose is achieved. The maximum dose that can be administered is 50 units daily, thus alternative therapy options should be considered if patients are anticipated to require more than 50 units. As with liraglutide monotherapy, if more than three days elapse without a dose of the combination product, reinitiate at 16 units daily and re-titrate.28
In addition to hypoglycemia, adverse reactions commonly associated with the combination product are nasopharyngitis, headache, nausea, diarrhea, increased lipase, and upper respiratory tract infection.28
AMYLIN RECEPTOR AGONIST: PRAMLINTIDE
Pramlintide (Symlin) is an amylin receptor agonist that has been approved for adjunctive treatment in patients with type 1 diabetes (T1D) or T2D who have failed to achieve glucose control despite optimal insulin therapy, including mealtime insulin.29 Amylin is a peptide that is co-secreted with insulin. The normal physiological function of amylin is poorly understood; however, activation of the amylin receptor by pramlintide has been shown to slow gastric emptying, suppress glucagon secretion, and promote satiety.12
Pramlintide is available as a 1 mg/mL solution in two prefilled pen devices. A 1.5 mL pen (SymlinPen 60) delivers doses of 15 mcg, 30 mcg, 45 mcg, and 60 mcg. A 2.7 mL pen (SymlinPen 120) delivers doses of 60 mcg and 120 mcg. Before initiation of pramlintide, mealtime insulin doses should be reduced by 50% to reduce the risk of hypoglycemia. Patients with T1D should start with 15 mcg injected immediately prior to each meal. The dose may be increased in 15 mcg increments once the patient has not experienced nausea for 3 consecutive days up to a maximum dose of 60 mcg before each meal. The highest tolerated dose should be used, but if patients cannot tolerate 30 mcg, then pramlintide should be discontinued. Patients with T2D should start with 60 mcg injected immediately prior to each meal. The dose should be increased to 120 mcg before each meal once the patient has not experienced significant nausea for 3 consecutive days. If 120 mcg is not tolerated, the 60-mcg dose should be used. If the patient skips a meal or is planning to eat a meal with less than 250 calories or less than 30 grams of carbohydrate, that dose of pramlintide should be skipped. After the initial 50% dose reduction, insulin doses should be re-titrated based on glycemic control and tolerability.29
Like GLP-1 RAs, pramlintide slows gastric emptying, which may affect the absorption of oral medications. The manufacturer does not list any major drug interactions but recommends that analgesics, antibiotics, and oral contraceptives be taken 1 hour before or 2 hours after taking pramlintide as a precaution.29
The most common adverse effects associated with pramlintide are nausea, vomiting, anorexia, and headache. Patients typically develop tolerance to the gastrointestinal side effects over time. Pramlintide is contraindicated for use in patients with hypoglycemia unawareness or confirmed gastroparesis. Pramlintide contains a black box warning because of the risk for severe hypoglycemia, particularly in patients with type 1 diabetes.29
As a result of the combination of minimal A1C reduction, frequent injections, and the high risk of hypoglycemia, this agent is not commonly used in practice. Careful assessment is needed to ensure pramlintide is appropriate for a given patient. Most importantly, it is crucial to determine that the patient is not at high risk for hypoglycemia and that the patient is able to effectively detect and treat hypoglycemia before it becomes severe.
CONCLUSION
Insulin therapy is not the only injectable medication for the management of diabetes. This module reviewed the non-insulin injectable medication classes available on the market today: GLP-1 RAs and amylin receptor agonists. GLP-1 RAs are given high recommendations for use in both the ADA and AACE/ACA treatment guidelines.2,3
Both GLP-1 RAs and amylin receptor agonists act to slow gastric emptying, suppress glucagon release, and promote satiety.6,20 In addition, GLP-1 RAs stimulate the release of insulin.6 Cardiovascular outcomes trials to date have been mixed, and liraglutide is the only medication in this class that currently carries an indication for cardiovascular risk reduction. All of these agents slow gastric emptying, which commonly causes gastrointestinal adverse effects and may interfere with the absorption of oral medications.16-22,29
As always, the selection of an appropriate medication requires assessing the patient as a whole. One should always consider the potential medication's place in therapy, adverse effects, contraindications, drug interactions, and ease of administration as well as the patient's comorbidities, A1C, and willingness and ability to use an injectable medication. While there are some differences among the various non-insulin injectables with regard to frequency of dosing and side effects, the main determining factor for selection will likely be cost and insurance coverage. The pharmacist plays a crucial role in ensuring that a non-insulin injectable is appropriate for the patient, recommending dosing changes based on tolerability and glycemic control, and ensuring patients understand both what to expect from therapy and how to administer the medications appropriately. The pharmacy technician plays an important role in understanding their place in therapy, pharmacology and reviewing side effects along with understanding when to refer the patient to the pharmacist for counseling.
Potential Counseling Tips for Pharmacists | Pharmacy Technician Referral Tips
|
| Category |
Potential Counseling Tips |
| Evaluating if Patient Is a Candidate for Injectable Medications |
Provide patient-centered counseling to assess and address attitudes toward needles and self-administered injections
Ensure patient understands the appropriate way to use the provided administration devices
Ensure patient is physically able to operate the device, including setting the dose, mixing, and injecting
Discuss options with patients that will increase ease of use, decrease dosing frequency, and reduce out-of-pocket costs |
| General Diabetes Education |
Remember that medications are not the only solution to diabetes management; lifestyle modification is key to managing diabetes. These topics should always be addressed during counseling.
Pharmacy Technicians can play a key part in upfront discussions with patients on their current lifestyle including diet, exercise, side effects and other medications. If patients have any questions in relation to management of their lifestyle, the patient should be referred to discuss with the pharmacists.
|
|
The Pharmacist's Role in Diabetes Care
The Pharmacy Technician's Role in Diabetes Care
|
To best assist a patient or prescriber in selecting diabetes therapies, consider national treatment guidelines, potential adverse effects, contraindications and precautions, drug interactions, and ease of use as well as specific comorbidities
Maintain a current, complete medication list for the patient and be aware of all of the patient's relevant conditions |
| Medication Tips for Pharmacists and Pharmacy Technicians |
The most common side effects of GLP-1 RAs as a class are nausea, diarrhea, and headache
Side effects are often experienced when starting the medication and may cease over time
Side effects may be reduced by lowering the dose and slowly titrating to the recommended dose
Nausea may be reduced by eating smaller portions and reducing fat intake |
REFERENCES
- Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017.Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017.
- American Diabetes Association. Standards of medical care in diabetes—2019. Diabetes Care. 2019;42(Suppl. 1):S1-S193.
- Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2019 executive summary. Endocr Pract. 2019;25(1):69-100.
- AstraZeneca Pharmceuticals. Pen user manual: Byetta exenatide injection. Retrieved from https://www.azpicentral.com/byetta/ifu_byetta.pdf. Accessed April 10, 2019.
- AstraZeneca Pharmaceuticals. Instructions for use: Bydureon. Retrieved from https://www.azpicentral.com/bydureon/ifu_bydureon.pdf. Accessed April 10, 2019.
- AstraZeneca Pharmaceuticals. Instructions for use: Once-weekly Bydureon BCise. Retrieved from http://www.azpicentral.com/bydureon_bcise/bydureon_bcise_ifu.pdf. Accessed April 10, 2019.
- Novo Nordisk. Victoza (liraglutide) instructions for use. Retrieved from http://www.novo-pi.com/victoza.pdf#patient. Accessed April 10, 2019.
- Eli Lilly and Company. Instructions for Use: Trulicity (dulaglutide). Retrieved from http://pi.lilly.com/us/trulicity-lowdose-ai-ifu.pdf. Accessed April 10, 2019.
- Novo Nordisk. Ozempic (semaglutide) instructions for use. Retrieved from http://www.novo-pi.com/ozempic.pdf#guide. Accessed April 10, 2019.
- Novo Nordisk. Xultophy 100/3.6 (insulin degludec and liraglutide injection) instructions for use. Retrieved from http://www.novo-pi.com/xultophy10036.pdf#IFU. Accessed April 10, 2019.
- Sanofi-aventis. Instructions for use: Soliqua 100/33. Retrieved from https://www.soliqua100-33.com/-/media/EMS/Conditions/Diabetes/Brands/soliqua-100-33/Global/IFU/soliqua100-33.pdf. Accessed April 10, 2019.
- Powers AC. Diabetes mellitus: Diagnosis, classification, and pathophysiology. In: Kasper D, Fauci A, Hauser S, Longo D, Jameson J, Loscalzo J, eds. Harrison's principles of internal medicine. 19th ed. New York, NY: McGraw-Hill; 2015. http://accessmedicine.mhmedical.com/content.aspx?bookid=1130&Sectionid=79752868. Accessed April 10, 2019.
- Powers AC. Diabetes mellitus: Management and therapies. In: Kasper D, Fauci A, Hauser S, Longo D, Jameson J, Loscalzo J, eds. Harrison's principles of internal medicine. 19th ed. New York, NY: McGraw-Hill; 2015. http://accessmedicine.mhmedical.com/content.aspx?bookid=1130&Sectionid=79752952. Accessed April 10, 2019.
- Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8(12):728-742.
- O'Mara NB. PL detail-document, drugs for type 2 diabetes. 2015.
- Byetta [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2018.
- Bydureon [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2019.
- Bydureon BCise [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2019.
- Victoza [package insert]. Plainsboro, NJ: Novo Nordisk, Inc.; 2017.
- Trulicity [package insert]. Indianapolis, IN: Eli Lilly and Company; 2019.
- Adlyxin [package insert]. Bridgewater, NJ: sanofi-aventis U.S. LLC; 2019.
- Ozempic [package insert]. Plainsboro, NJ: Novo Nordisk, Inc; 2017.
- Wysham CH, MacConell LA, Maggs DG, et al. Five-year efficacy and safety data of exenatide once weekly: long-term results from the DURATION-1 randomized clinical trial. Mayo Clin Proc. 2015;90(3):356-365.
- Saxenda [package insert]. Plainsboro, NJ: Novo Nordisk, Inc.; 2018.
- Sanofi-aventis. Instructions for use: Adlyxin. Retrieved from http://products.sanofi.us/Adlyxin/Adlyxin.pdf. Accessed May 13, 2018.
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
- Soliqua 100/33 [package insert]. Bridgewater, NJ: sanofi-aventis U.S. LLC; 2019.
- Xultophy 100/3.6 [package insert]. Plainsboro, NJ: Novo Nordisk, Inc.; 2019.
- Symlin [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2016.
Back to Top