
Expired activity
Please go to the PowerPak
homepage and select a course.
Emergency Contraception: An Update for Pharmacists
INTRODUCTION
Of the 72.2 million reproductive age women (15-49 years) in the United States, approximately 65% were using contraception.1 This leaves many women vulnerable to unintended pregnancy. In 2011, 45% of the 6.1 million pregnancies were unintended.2 Yet only 5% of unintended pregnancies occur when contraception is used correctly and consistently.3 Furthermore, situations such as sexual assault or reproductive coercion can lead to unintended pregnancy. Consequences of unintended pregnancy include abortion and public expenditures of $21 billion.4 Therefore, preventing unintended pregnancy is critical from individual, public health, and economic perspectives.
It is known that 60% of all women of reproductive age currently use a contraceptive method, mostly to prevent pregnancy. 3 Common methods include the oral contraceptive pill (15.6%); female sterilization (13.4%); male condoms (8.9%); intrauterine devices (IUDs) (7.2%); injectables (2.4%); vaginal rings (1.5%); implants (1.6%); and contraceptive patches (0.1%).3 Some women may not know that the probability of pregnancy after intercourse on any given day of the month varies, but the probability reaches its peak (9.7%) on menstrual day 13 and remains high through menstrual day 22.5 Since most contraceptive users choose methods other than female sterilization or vasectomy (which are permanent), or IUDs or implants (which are long acting), correct and consistent use is critical for maximal effectiveness. However, methods such as oral contraception, vaginal rings, or condoms require user attention and daily, monthly, or “just in time” adherence, respectively. The typical user may experience higher failure rates (ie, pregnancy) with these methods. For example, the typical failure rate for oral contraceptives or vaginal ring is 9% and the typical failure rate for male condoms is 13%; in contrast, IUDs and implants have failure rates lower than 1%.3
Unprotected sex and/or contraceptive failure occur for many and often complex reasons:6
- sexual assault or reproductive coercion
- lapse in contraceptive adherence (eg, missed dose)
- contraceptive mishap (eg, condom break, IUD expulsion, diaphragm dislodged, spermicide not inserted prior to sex)
- reduced contraceptive effectiveness (eg, drug interactions)
- lack of contraceptive use
Emergency contraception (EC), or postcoital contraception, offers an opportunity to prevent pregnancy when unprotected or inadequately protected sex occurs. In the United States, 3 types of emergency contraceptive pills (ECPs) and the copper IUD are available for EC.
EC’s rapidly evolving regulatory and clinical landscape emphasizes the importance of health care providers having accurate, updated knowledge and skills to provide optimal patient care. Several organizations have published guidelines, recommendations, or position statements to help health care providers provide appropriate EC.7-11 This article updates pharmacists on the current clinical and regulatory information related to EC and describes effective strategies for educating consumers about its use.
METHODS OF EMERGENCY CONTRACEPTION
The primary forms of EC include the copper IUD, combined ECPs containing both estrogen and progestin, progestin‐only ECPs, and ECPs containing an antiprogestin (either mifepristone or ulipristal acetate [UPA]). Each of these will be discussed individually in greater detail, but it is important to discuss comparative effectiveness first. Given that EC is a preventive therapy, effectiveness is best measured by comparing the probability that the condition (pregnancy) will occur if the therapy is used to the probability that it will occur without treatment.12 For the most accurate estimates, statisticians estimate the chance that pregnancy would occur in the absence of EC using published data on the probability of pregnancy on each day of the menstrual cycle. Then, they compare that data to the actual number of pregnancies observed after treatment in trials.12 Effectiveness is calculated as 1–O/E, where O and E are the observed and expected number of pregnancies, respectively.12 While this is currently the most accurate method to calculate effectiveness, not all estimates of effectiveness have been calculated this way. This has caused wide variations and imprecise numbers to be reported in effectiveness estimates.
Copper Intrauterine Device
Effectiveness
Numerous studies have demonstrated that the copper IUD is the most effective form of EC, and insertion within 5 days of unprotected sex is most often recommended.12-16 In a review of 42 studies that enrolled 7034 women receiving an IUD within 2 to10 days (majority within 5 days), the pregnancy rate was 0.09%.17 In a study of 542 women receiving either copper IUD or oral levonorgestrel (LNG) EC, the 1-year cumulative pregnancy rate was 5.2% for copper IUD users vs 12.3% for oral LNG users (HR 0.42, 95% CI: 0.20–0.85, P = 0.017).18 When controlling for demographic variables, women who chose the IUD for EC had fewer pregnancies and were half as likely to be pregnant in the following year compared with those choosing oral LNG (HR 0.50, 95% CI: 0.26–0.96, P = 0.037).
Mechanism of action
The copper IUD appears to have pre- and post-fertilization contraceptive effects. The copper IUD releases copper ions to enhance the inflammatory response and reach copper concentrations in the genital tract that are toxic for spermatozoa.19 Prior to fertilization, copper may inhibit sperm migration and viability at the level of the cervix, endometrium, and fallopian tube to prevent fertilization; slow (but not inhibit) the transport of the ovum through the fallopian tube; and damage or destroy the ovum.20 The IUD’s possible post-fertilization mechanisms of action include slowing or speeding the early embryo’s transport through the fallopian tube; damage to or destruction of the early embryo before it reaches the uterus; and prevention of implantation. Therefore, the copper IUD works through several different potential mechanisms and can be inserted up to 120 hours after unprotected intercourse.
Contraindications
According to the Centers for Disease Control and Prevention (CDC) medical eligibility criteria, the following conditions are considered contraindicated or category 4 (unacceptable health risk if contraceptive method is used) for insertion: pregnancy, recent history of puerperal sepsis in postpartum women, unexplained vaginal bleeding, gestational trophoblastic disease with elevated beta-human chorionic gonadotropin levels or malignant disease, distorted uterine cavity, endometrial cancer, pelvic tuberculosis, cervical cancer awaiting treatment, postpartum sepsis, immediate postseptic abortion, current pelvic inflammatory disease (PID), and current purulent cervicitis or chlamydial infection or gonorrhea.21 Women with Wilson’s disease or hypersensitivity to anything in the copper IUD should not receive it.22 When the copper IUD is the only EC option available, prophylactic antibiotics may be considered if the clinician suspects the patient has an infection, or the country in which insertion will take place has a high prevalence of cervical gonococcal and chlamydial infections and limited access to infection screening.23 Women with a history of PID or sexually transmitted infections (STIs) can safely receive the copper IUD.
Dosing
The copper T 380 A (Paragard) is a T-shaped IUD that contains polyethylene with copper along the vertical stem and horizontal arms. It is approved for up to 10 years of use; however, studies have shown it to be effective for longer periods of time and at least 12 years.24,25 This provides an added benefit of extended contraception beyond its use for EC. The CDC, World Health Organization (WHO), and the United Kingdom Faculty of Sexual & Reproductive Healthcare indicate that an IUD can be used beyond 5 days after unprotected intercourse if the time of ovulation is known.8 Otherwise, the insertion should not occur more than 5 days after unprotected intercourse to ensure that the device is inserted before embryo implantation (and ensuring its contraceptive function rather than as a potential pregnancy termination modality).26 While the uterine cavity in nulliparous women tends to be smaller than parous women, trained professionals can insert copper IUDs safely into women who have not had children.27,28
Potential drug interactions
There are no reported drug interactions with the copper IUD.
Adverse effects
The copper IUD’s adverse effects are similar whether used for contraception or EC.29 Pain and/or discomfort are common adverse effects related to IUD insertion; however, the risk of uterine perforation is very low (1/1000 insertions).30 Risk of malposition and expulsion is also low but may potentially occur.31 No cases of PID were detected in 2 studies of 2160 women using copper IUD for EC.32 Perhaps the most problematic issues with using the copper IUD as EC are time and accessibility since a trained health care provider must perform the procedure.Cost may also be a concern as out-of-pocket cost for an IUD insertion can be approximately $975 (a one-time cost); the average cost of oral EC regimens is typically $40 to $50.33-35 Insurance companies will typically cover IUDs and oral EC regimens when prescribed for contraception with zero copayment or deductible (but may not reimburse for over-the-counter EC). If the copper IUD is continued for contraception, menstrual cycle bleeding can become longer and heavier with an increased likelihood of dysmenorrhea.36 During copper IUD use, menstrual blood loss increases by approximately 50%, which may lead to iron depletion anemia.37
Ulipristal Acetate (Ella)
Effectiveness
Ulipristal acetate (UPA) is more effective than LNG for EC and remains effective for up to 5 days.12,13 A meta-analysis of 2 trials comparing UPA and LNG demonstrated that UPA was significantly more effective than LNG, but the absolute difference was small (22/1617 pregnancies [1.4%] vs 35/1625 pregnancies [2.2%]; OR 0.58, 95% CI 0.33–0.99).38,39 In this analysis, UPA’s failure rate was lower than LNG’s. The odds ratio for pregnancy was 65% lower in the first 24 hours, 42% lower up to 72 hours, and 45% lower up to 120 hours.39 UPA’s efficacy does not appear to decrease in the 120 hours after unprotected sex; LNG has decreasing effectiveness over time.38-40
This efficacy pattern appears to occur because UPA is more effective than LNG at delaying ovulation. With LNG, when the leading follicle reaches 15 to 17 mm, ovulation is prevented similarly to placebo. With UPA, when the leading follicle reaches 18–20 mm (indicating ovulation should occur within 48 hours) and the probability of conception exceeds 30%, UPA prevents follicular rupture within 5 days of administration in 59% of cycles, compared with 0% in placebo cycles.41,42 Mifepristone and UPA probably have similar efficacy as contraceptives.
Mechanism of Action
UPA is a second-generation selective progesterone receptor modulator (SPRM) that acts as an antagonist in progesterone-responsive tissues and binds to progesterone receptors with high affinity.43-46 With its antiprogestin properties, it delays ovulation and prevents implantation effectively for up to 120 hours after unprotected intercourse. Administered immediately prior to ovulation, 30 mg UPA delays ovulation up to 5 days.47 The ability of UPA to delay ovulation depends on luteinizing hormone (LH) levels with complete effectiveness before the LH surge, 78.6% prior to the LH peak, and 8.3% after the LH peak. Even when given just prior to ovulation, UPA blocks or significantly delays follicular rupture for more than 5 days in 59% of women.48
Contraindications
There are no contraindications to UPA except known or suspected pregnancy.49
Dosing
The approved dose of UPA for EC is 30 mg orally once. UPA requires a prescription in the United States (it has nonprescription status in Europe). Women should take the tablet as soon as possible and within 120 hours of unprotected intercourse. Women should also start or use another form of contraception before another act of intercourse occurs. If a hormonal method (eg, oral contraceptives, vaginal ring, implants, injection) will be initiated for routine contraception, women should use a barrier method until menstruation occurs, and then start the hormonal method after the menstrual period.6 Initiating or returning to hormonal contraception using a “quick start” method is of concern; competitive binding at the progesterone receptor may lead to ovulation later in the menstrual cycle and increase the possibility of a late-cycle pregnancy. Before insertion of a progestin-only implant or LNG-releasing intrauterine system, pregnancy should be ruled out.
Potential Drug Interactions
Due to the action on progesterone receptors, the potential interaction with combined hormonal contraceptives (estrogen and progestin) and progestin-only contraceptives warrants consideration.6,50 No data demonstrate interference with hormonal contraception, but the efficacy of both UPA and the contraceptive containing progestin could be reduced. The use of barrier methods is advisable for 14 days or until the next menstruation.50 Enzyme inducers, specifically those that induce cytochrome P450 3A4 (CYP3A4), may reduce UPA’s effectiveness by increasing catabolism and decreasing serum concentrations.49,51 Patients should avoid barbiturates, bosentan, carbamazepine, felbamate, griseofulvin, oxcarbazepine, phenytoin, rifampin, St. John’s Wort, and topiramate.49 Inhibitors of CYP3A4, such as itraconazole or ketoconazole, increase UPA plasma concentrations.
Adverse Effects
UPA’s adverse effects are similar to that of LNG and include headache (19%), dysmenorrhea (13%–14%), nausea (11%–13%), and menstrual disturbances (24% or greater). Women taking UPA may experience menstruation 2 days later than expected.39 Based on limited data, UPA exposure in pregnancy does not appear teratogenic.52 In postmarketing surveillance at 1 year, a total of 553 women experienced 1049 adverse drug reactions. The most frequent were pregnancies (282, 6.8%); nausea, abdominal pain, and vomiting (139, 13.3%); headache and dizziness (67, 6.4%); and metrorrhagia (bleeding at irregular intervals), menses delay, and breast symptoms (84, 8.0%).52
One recent controversy is the potential effect of body weight on the efficacy of oral ECPs. Two trials comparing UPA and LNG were combined into an analysis, and 1731 women taking LNG showed a meaningful increase in pregnancy rate with increased weight (1.5% for those weighing less than 65 kg and 6% for those weighing more than 75 kg). Women who weighed 75 kg or more had similar pregnancy rates to women who did not use EC.53 Some data indicate all oral EC are significantly less effective in obese women (ie, BMI exceeding 30), with pregnancy rates 3 times higher compared with nonobese women.54 In women with BMI of 26, pregnancy rates were no different for LNG users compared to nonusers; UPA appears to lose effectiveness at a BMI higher than 35.54-56 Overall, available data appear limited and cannot definitively conclude that EC efficacy is reduced with increased weight.57 The European Society on Contraception published a consensus opinion paper on contraception in obese women.58 The summary recommendation indicates that for obese women at high risk for pregnancy after unprotected intercourse, the copper IUD is the preferred method. If that is not possible, UPA 30 mg may be a better option than LNG 1.5 mg. Nearly half of all women using contraceptives are overweight or obese, so this issue is important to research further.59 Until more evidence appears, all overweight and obese women should be offered all forms of EC; however, the copper IUD would certainly be the most effective.
Levonorgestrel-Only Emergency Contraception
Effectiveness
As stated above, LNG is not as effective as the copper IUD or UPA for EC. LNG is more effective than the Yuzpe regimen, the prototypical EC method discussed below (RR 0.57, 95% CI 0.39–0.84).13 LNG’s efficacy decreases with increasing time since unprotected intercourse. Its efficacy is estimated to be 79% if taken within 72 hours and 60% if taken within 72 to 120 hours.60 In a multinational trial coordinated by WHO, LNG’s failure rate was 1.1% compared with the Yuzpe regimen at 3.2%.61
Mechanism of Action
LNG’s primary EC mechanism of action is ovulation delay or inhibition.62 It has been shown that administering LNG after the onset of the LH surge was ineffective in inhibiting ovulation and it does not prevent embryo implantation.63,64 Some studies indicate, therefore, that LNG is effective as EC only when administered before ovulation. The exact time of ovulation cannot be easily determined, and LNG has a low adverse event profile; therefore, LNG can and should be offered to women seeking EC at any time.
Contraindications
There are no contraindications to LNG EC except current pregnancy.
Dosing
The initial approved dosing of LNG EC was 2 of the 0.75 mg tablets taken 12 hours apart, but now has been simplified to a single dose of 1.5 mg. It is available over the counter (OTC). It is effective for up to 5 days after unprotected intercourse but is approved for use within 72 hours. It is more effective the sooner it is taken. Women should start or use a routine form of contraception before another act of intercourse occurs. Women who choose a hormonal method (eg, oral contraceptives, vaginal ring, implants, injection) should start the same day as the LNG regimen or the following day, but also use a barrier method for the first 7 days. Alternatively, women can start a hormonal contraception after the next menstrual period with use of a barrier method until menstruation occurs.6 Before insertion of a progestin-only implant or LNG-releasing intrauterine system, pregnancy should be ruled out.
Potential Drug Interactions
No data are available regarding drug interactions with oral EC, but is clinically reasonable to assume agents that can reduce other hormonal contraceptives’ efficacy may also affect oral EC’s efficacy. Similar to UPA, the product information states that progestin-metabolizing enzymes, including CYP3A4, may decrease the plasma concentrations of progestins, and decrease the effectiveness of LNG EC. Similar drugs or herbal products listed in the UPA section may decrease oral LNG’s effectiveness. Additionally, significant changes (increase or decrease) in LNG plasma levels have been noted with coadministration of HIV protease inhibitors or non-nucleoside reverse transcriptase inhibitors (NNRTIs).
It is not known if LNG EC’s efficacy is reduced by recent use of UPA (a progesterone receptor modulator). Therefore, if a woman has recently used a LNG EC regimen and has a subsequent need for EC, clinicians should recommend the LNG regimen or copper IUD rather than UPA.
Adverse Effects
When compared with the Yuzpe regimen, LNG causes 50% less nausea and 70% less vomiting.60,65 Women taking LNG may menstruate 1 day earlier than expected.60 Additional studies suggest that other adverse effects of combined hormonal contraception are reduced including dizziness, headache, asthenia, breast tenderness, and abdominal pain.66 Two studies of 357 women (total) have shown no significant differences in pregnancy outcomes for those exposed to LNG taken during a conception cycle compared to those unexposed.67,68 Furthermore, one study with a 2-year follow-up demonstrated no difference in children’s physical or mental development based on exposure.69 When LNG fails, ectopic pregnancy is no more likely than in the general population.70 The controversy regarding EC efficacy and body weight (including LNG) was discussed in adverse effects related to UPA.
Combined Oral Contraceptives (Yuzpe)
Efficacy
Invented in 1974 by Dr Albert Yuzpe, this EC regimen employs oral combined hormonal contraceptives containing estrogen (ethinyl estradiol 100 mcg) and progestogen (LNG 0.5 mg). As the time between unprotected sex and treatment lengthens, the Yuzpe regimen’s efficacy decreases; the failure rate ranges from 2% to 3.5% when administered within 72 hours of unprotected sex.61,71,72 As an estimate, combined hormonal contraceptives used as EC reduce the risk of pregnancy by approximately 75%.73 This method is the least effective of all EC methods.
Mechanisms of Action
The Yuzpe regimen works similarly to routine hormonal contraception in that it inhibits or delays ovulation.74 However, when used as EC, clinical studies have not definitively demonstrated that combined hormonal contraceptives alter the endometrium or interfere with the function of the corpus luteum; thicken cervical mucus; alter the tubal transport of sperm, egg, or embryo; or inhibit fertilization.
Contraindications
Only current pregnancy is an absolute contraindication for the Yuzpe regimen. Risks associated with a single course of combined hormonal contraceptives are considered negligible.
Dosing
This regimen is typically provided off-label using several combined oral contraceptive pills to provide the necessary dose. The woman takes the first dose as soon as possible and the second dose 12 hours later. Each dose should contain at least 100 mcg of estrogen (usually ethinyl estradiol) and 0.5 mg of progestin, so the number of pills will depend on which daily oral contraceptive the woman uses. Women should start the regimen within 72 hours of unprotected sex, but can use it within 120 hours.
Drug Interactions
Drugs that may impact efficacy of combined hormonal contraceptives may impact the efficacy of the Yuzpe regimen; another EC method (eg, copper IUD) should be considered if the interaction is significant.
Adverse Effects
Approximately 50% and 20% of Yuzpe regimen users experience nausea and vomiting, respectively.61,75 These adverse effects primarily result from the combined hormonal contraceptive regimens’ high estrogen dose. Some studies have shown that preventive use of antinausea medication 1 hour prior to EC tablet administration can help minimize nausea by 27% and vomiting by 64%, but routine use is not always supported as evidence is limited to expert opinion and package labeling.61,76,77 Women may plan to take dose near bedtime to minimize the risk of nausea and vomiting. Women should repeat the dose if they vomit within 1 hour of taking a dose of any type of EC.78
Given the high dose of estrogen that is typically consumed with the Yuzpe regimen, there is a risk (albeit small) that thromboembolism could occur. This is a well-known adverse effect in long-term use of combined hormonal contraception; however, it must be thought of within the context of pregnancy prevention since risk of thromboembolism is much higher in pregnancy.79
Mifepristone (Low Dose)
Mifepristone is a first generation SPRM with high efficacy as EC, but it is not available to pharmacists in the United States for any purpose at this time. At doses used for EC, mifepristone can prevent implantation.80 The efficacy of low-dose mifepristone (doses lower than 25 mg) is comparable or greater than LNG for EC (RR 0.72, 95% CI 0.52–0.99).13 When comparing mifepristone 5 mg and 10 mg within 6 days of an unprotected sex act in 2418 women seeking EC, 88% and 93% of pregnancies were prevented, respectively.81 Delayed menstruation by 7 days or more was experienced by 4.9% and 11% in the 5 mg and 10 mg groups, respectively (P = 0.001). There was a significant high failure rate (RR 5.1, 95% CI 1.6–16.4) for women weighing more than 75 kg in the 5 mg group, making mifepristone 10 mg the more appropriate dose for EC. Ninety-one percent of women in the 5 mg group and 90.9% of women in the 10 mg group reported no adverse effects. Eight women reported vaginal bleeding.
ACCESS TO EMERGENCY CONTRACEPTION
Prescription Status (OTC/Rx)
Currently, FDA has approved 4 types of EC products. These include brand named EC products (eg, Plan B One-Step, Take Action, and Aftera), other generic one-pill LNG EC products, and UPA. Currently, LNG EC including Plan B One-Step and its generic form Take Action, Next Choice One Dose, My Way, or other generics are approved for OTC sale without age restrictions. Due to its high cost, some pharmacies may keep OTC EC behind the pharmacy counter to protect against theft; however, pharmacies should make every effort to make it easily accessible. Oral UPA remains available by prescription only, although in some states, pharmacists may dispense it under collaborative practice agreements or state-approved protocols.
Since the late 1990s, different states have taken various approaches to expand EC access. Some differences include EC in hospital emergency departments, pharmacy protocols, and restrictions. Table 1 outlines highlights for states with specific EC policies.82 Some states have gone beyond EC issues and developed expanded contraception protocols. For example, 8 states allow pharmacists to dispense EC without a physician’s prescription under certain conditions. California laws allow pharmacists to dispense EC without prescription under collaborative practice agreement or state-approved protocols. Other states such as Hawaii, Maine, Massachusetts, New Hampshire, and New Mexico have also enacted these types of laws.82
Table 1. State EMERGENCY CONTRACEPTION POLICIES
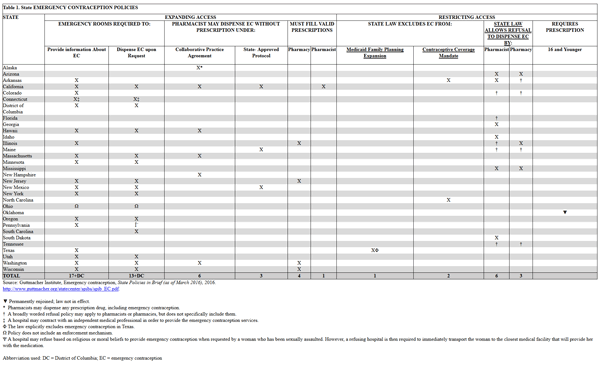
please click on the image for a larger view.
Advanced Provision of EC
Advanced provision is the practice of supplying oral EC in advance to ensure that EC will be available when needed and can be taken as soon as possible after unprotected intercourse. The context of advanced provision has shifted slightly as some EC products gained OTC status. Advanced provision remains an important concept for research; findings would have public health implications if studies determine that having EC on hand when needed reduces unintended pregnancy rates. Advanced provision is especially important for pharmacists to understand as it provides an opportunity to perform patient education about pregnancy risks and appropriate use of EC (eg, timing, dose).
Some information suggests that increasing the availability of EC is unlikely to yield a significant decrease in unintended pregnancy. A Cochrane review of 11 studies representing 7695 patients showed no decrease in pregnancy rates for studies with 12-month follow-up (OR 0.98; 95% CI 0.76–1.25), 7-month follow-up (OR 0.48; 95% CI 0.18–1.29), 6-month follow-up (OR 0.92, 95% CI 0.70–1.20) and 3-month follow-up (OR 0.49, 95% CI 0.09–2.74). In this analysis, advance provision did not increase rates of STIs, increase frequency of unprotected intercourse, or result in changes in contraceptive methods. Similar findings were seen in a systematic review of 17 studies.83 In another analysis of 74 women given immediate, free access to EC, women had more frequent sex or substituted EC for other more effective methods.84 Randomized trials and systematic reviews have demonstrated no increase in unintended pregnancy or abortion with EC, but data regarding incidence of STIs with increased access to EC is conflicting.85-92 Differences in state and federal policies may influence sexual practices, including higher rates of unprotected sex in states with their own policy changes.
COUNSELING ABOUT EMERGENCY CONTRACEPTION
While up to 86% of adolescent women have heard of EC, many incorrectly identify how it works, how it can be obtained, or when it can be administered.93-95 Studies emphasize the importance of the pharmacist’s ability to communicate effectively with patients about appropriate EC use. When properly counseled, patients have positive attitudes and high satisfaction with the EC information they receive.96
A tiered approach to EC counseling has been suggested to improve knowledge and provide information in a way that patients prefer.97,98 This approach encourages providers to present information about efficacy in categories or tiers for typical use (eg, order of effectiveness), even when all options are not available at the provider’s location. Providers can also discuss when EC needs to be used, how to use EC, and where it can be obtained with patients.98 EC is not intended to be a routine form of contraception because many more effective and affordable methods are available. There are no long-term data about the efficacy or safety of various EC regimens when used frequently or repeatedly over long periods of time. A systematic review of 15 studies found that LNG 0.75 mg given multiple times per cycle caused no serious adverse events.99 This provides reassurance that using LNG EC as often as needed is safe. Repeated use of UPA has not been studied at the EC dose (30 mg) but administering 5 mg to 10 mg for several weeks to treat uterine fibroids appears safe and well-tolerated.100 The product information for UPA EC recommends against using the product more than once in the same cycle. If subsequent EC is needed, a copper IUD is the preferred form of EC. If an IUD is not possible, LNG EC may be acceptable.
Additional unprotected sex soon after oral EC is associated with pregnancy risk. Therefore, pharmacists need to provide information about the importance of initiation or continuation of routine (daily, weekly, monthly, or longer) contraception, which is more effective than EC. Immediate initiation of hormonal contraception is not indicated following UPA, as this may lead to unintended pregnancies; however, nonhormonal or barrier contraceptive methods can be used. In a managed care organization model, adolescents and women who accessed EC through a call center were less likely to become pregnant within 3 months than women who accessed it through office visits (adjusted OR 0.82, 95% CI 0.72–0.94); however, they were more likely to become pregnant within 4 to 6 months (adjusted OR 1.37, 95% CI 1.16–1.60). Directly providing combined hormonal contraceptives (where allowed by protocol) with EC or offering rapid access to a family-planning clinic significantly increases the use of effective ongoing contraception.101 Additional strategies, including simplifying access to IUDs, are needed to increase the use of effective contraception after EC. The opportunity for same-day placement of IUDs has been shown to increase knowledge and use of IUDs 3 months after EC was sought.102
Pharmacists have the unique advantage of knowing all forms of contraception (eg, barrier methods) a patient is using on a routine basis. Pharmacists should remind patients using short-term or user-dependent methods that EC is available and an important form of backup contraception.
Women (and men) who come to the pharmacy for EC are often distressed and concerned about potential pregnancy. It is important for pharmacists to demonstrate empathy while helping patients understand their options with expected efficacy, potential adverse effects, and timing of administration. Pharmacists can help screen for contraindications and offer information about what to expect from the EC method chosen. Table 2 provides sample open-ended questions and conversation techniques for a patient requesting LNG EC.
| Table 2. Approach to Patient Counseling for LNG EC |
| Question: What are you taking this medication for? |
Pharmacist: Verify understanding or appropriate indication.
- This medication is used to prevent pregnancy when unprotected or inadequately protected sex occurs.
- Screen for contraindications (if applicable).
|
| Question: How do you plan to take this medication? |
Pharmacist: Assess patient knowledge of using EC and describe dose/duration.
- Example: Have you ever taken EC before? When will you take this dose? What do you plan to do for contraception once this medication has been taken?
- This single tablet (1.5 mg) dose should be taken as soon as possible and no later than120 hours after the unprotected intercourse occurred (within 72 hours is the approved and preferred time frame).
- A routine form contraception should be instituted before another act of intercourse occurs.
- Assuming missed dose(s) of combined hormonal contraceptive pills, advise patient to continue pills as directed starting that day or the following day; patients should use additional barrier method contraception (eg, condom) for 7 days.
|
| Question: What beneficial and adverse effects may occur with this medication? |
Pharmacist: Describe patient goals or benefits of therapy and relevant adverse effects or precautions.
- This medication will help prevent pregnancy and has been shown to reduce pregnancy rates by 79%. This is an estimate based on the number of pregnancies that would have occurred if EC had not been used.
- This is not the most effective form of EC. The copper IUD and ulipristal acetate are more effective; they require either insertion by a provider or a prescription, respectively (if the patient is obese, may need to emphasize efficacy and EC options).
- This medication is very well tolerated; adverse effects that have been reported include dizziness, headache, weakness, breast tenderness, and abdominal pain.
- This may affect your menstrual period by having it (on average) 1 day earlier. If your period is more than 1 week late, please see a provider for a pregnancy test.
- Please see a provider if you experience irregular bleeding, abdominal or pelvic pain, and dizziness.
|
| Question: Can you repeat back key information we discussed so I can be sure we covered the most important points? |
Pharmacist: Verify patient understanding.
- Listen for appropriate dose and frequency/timing, what to expect (adverse effects and menstrual cycle), and plans to proceed (or not) with routine contraception.
|
| Abbreviations: EC, emergency contraception; IUD, intrauterine device; LNG, oral levonorgestrel |
Just as it is important for pharmacists to help patients select EC, it is also important for pharmacists to recognize when it may be most appropriate to refer the patient to a provider. Although EC has not been shown to cause ectopic pregnancies, they can occur after a woman uses EC.103 Symptoms of an ectopic pregnancy include irregular bleeding, abdominal or pelvic pain, and dizziness. Pharmacists should urge patients to contact a health care provider immediately if any of these symptoms occur. They should refer women to a health care provider for pregnancy testing if menstruation is more than a week late.
CONCLUSION
Several safe, effective EC options are available to all women at risk of pregnancy following unprotected or inadequately protected sex. The copper IUD has pre- and post-fertilization effects and is the most effective form of EC when inserted by a trained professional within 5 days of unprotected sex. Hormonal methods, including UPA, LNG, and combined hormonal contraceptives work by delaying or inhibiting ovulation. Their effectiveness is time dependent, and they should be used as soon as possible and within 120 hours. Although clinical data are lacking, drug interactions with hormonal EC methods may exist and caution should be used with concurrent medications metabolized by the CYP3A4 system. UPA and LNG regimens are generally well tolerated; onset of the menstrual cycle may vary slightly and referral to a provider should occur if it is beyond 1 week from the expected date of the period.
The LNG EC ‘s OTC availability facilitates access and gives pharmacists an opportunity to discuss appropriate use of EC and routine forms of contraception. Patients may need to use barrier methods until they can start a routine form of contraception. Pharmacists should communicate information with patients in an empathetic and open manner to facilitate helpful dialogue. Effective communication about contraception may heighten awareness about EC and incentivize women to use EC in a timely fashion when indicated.
REFERENCES
- Daniels K, Abma J. Current contraceptive status among women aged 15–49: United States, 2015–2017. NCHS data brief, no 327. Hyattsville, MD: National Center for Health Statistics. 2018.
- Guttmacher Institute. Unintended pregnancy in the United States. January 2019. Available at: https://www.guttmacher.org/fact-sheet/unintended-pregnancy-united-states. Accessed June 22, 2019.
- Guttmacher Institute. Contraceptive use in the United States. July 2018. Available at: https://www.guttmacher.org/fact-sheet/contraceptive-use-united-states. Accessed June 22, 2019.
- Guttmacher Institute. Unintended pregnancies cost federal and state governments $21 billion in 2010. March 2015. Available at: https://www.guttmacher.org/news-release/2015/unintended-pregnancies-cost-federal-and-state-governments-21-billion-2010. Accessed June 22, 2019.
- Li D, Wilcox AJ, Dunson DB. Benchmark pregnancy rates and the assessment of post-coital contraceptives: an update. 2015;91:344-349.
- Raymond EG, Cleland K. Emergency contraception. N Engl J Med. 2015;372:1342-1348.
- International Consortium for Emergency Contracpetion. Emergency contraceptive pills: medical and service delivery guidelines guidance, fourth edition. 2018. Available at: https://www.cecinfo.org/icec-publications/emergency-contraception-pills-medical-service-delivery-guidelines-fourth-edition/. Accessed June 22, 2019.
- Faculty of Sexual and Reproductive Healthcare. FSRH guideline: emergency contraception. December 2017. Available at: https://www.fsrh.org/standards-and-guidance/current-clinical-guidance/emergency-contraception/. Accessed June 22, 2019.
- Practice Bulletin No. 152: Emergency contraception. Obstet Gynecol.2015;126:e1-e11.
- Glasier A, Gemzell-Danielsson K, Bouchard P, et al. ISGE statement on oral emergency contraception. Gynecol Endocrinol.2014;30:681-682.
- World Health Organization. Emergency contraception. February 2018. Available at: https://www.who.int/news-room/fact-sheets/detail/emergency-contraception. Accessed June 22, 2019.
- Trussell J, Raymond EG, Cleland K. Emergency contraception: a last chance to prevent unintended pregnancy. January 2019. Available at: http://ec.princeton.edu/questions/ec-review.pdf. Accessed June 22, 2019.
- Shen J, Che Y, Showell E, Chen K, Cheng L. Interventions for emergency contraception. Cochrane Database Syst Rev. 2019;1:CD001324. doi: 10.1002/14651858.CD001324.pub6.
- Batur P, Kransdorf LN, Casey PM. Emergency contraception. Mayo Clin Proc. 2016;91(6):802-807.
- Wu S, Godfrey EM, Wojdyla D, et al. Copper T380A intrauterine device for emergency contraception: a prospective, multicentre, cohort clinical trial. Br J Obstet Gynaecol.2010;117:1205-1210.
- Zhou L, Xiao B. Emergency contraception with Multiload Cu-375 SL IUD: a multicenter clinical trial. 2001;64:107-112.
- Cleland K, Zhu H, Goldstuck N, Cheng L, Trussell J. The efficacy of intrauterine devices for emergency contraception: a systematic review of 35 years of experience. Hum Reprod. 2012;27:1994-2000.
- Turok DK, Jacobson JC, Dermish AI, et al. Emergency contraception with a copper IUD or oral levonorgestrel: an observational study of 1-year pregnancy rates. 2014;89:222-228.
- Ortiz ME, Croxatto HB, Bardin CW. Mechanisms of action of intrauterine devices. Obstet Gynecol Surv.1996;51:S42-S51.
- Stanford JB, Mikolajczyk RT. Mechanisms of action of intrauterine devices: update and estimation of postfertilization effects. Am J Obstet Gynecol.2002;187:1699-1708.
- Curtis KM, Tepper NK, Jatlaoui TC, et al. U.S. medical eligibility criteria for contraceptive use, 2016. MMWR Recomm Rep. 2016;65(No. RR-3):1–104. DOI: http://dx.doi.org/10.15585/mmwr.rr6503a1
- Paragard®[package insert]. Sellersville, PA: Teva Women’s Health, Inc. 2013. Available at: http://paragard.com/Pdf/ParaGard-PI.pdf. Accessed September 4, 2015.
- Black KI. Developments and challenges in emergency contraception. Best Pract Res Clin Obstet Gynaecol. 2009;23:221-231.
- Long-term reversible contraception. Twelve years of experience with the TCu380A and TCu220C. 1997;56:341-352.
- Sivin I. Utility and drawbacks of continuous use of a copper T IUD for 20 years. 2007;75:S70-S75.
- Turok DK, Godfrey EM, Wojdyla D, Dermish A, Torres L, Wu SC. Copper T380 intrauterine device for emergency contraception: highly effective at any time in the menstrual cycle. Hum Reprod.2013;28:2672-2676.
- Kurz KH, Tadesse E, Haspels AA. In vivo measurements of uterine cavities in 795 women of fertile age. 1984;29:495-510.
- Otero-Flores JB, Guerrero-Carreno FJ, Vazquez-Estrada LA. A comparative randomized study of three different IUDs in nulliparous Mexican women. 2003;67:273-276.
- Glasier A. Emergency contraception: clinical outcomes. 2013;87:309-313.
- Heinemann K, Reed S, Moehner S, Minh TD. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. 2015;91:274-279.
- Aiken AR, Trussell J. Recent advances in contraception. F1000Prime Rep.2014;6:113.
- Murphy PA, Jacobson J, Turok DK. Criterion-based screening for sexually transmitted infection: sensitivity, specificity, and predictive values of commonly used questions. J Midwifery Womens Health.2012;57:622-628.
- Turok DK, Gurtcheff SE, Handley E, et al. A survey of women obtaining emergency contraception: are they interested in using the copper IUD? 2011;83:441-446.
- Trussell J, Hassan F, Lowin J, Law A, Filonenko A. Achieving cost-neutrality with long-acting reversible contraceptive methods. 2015;91:49-56.
- American Society for Emergency Contraception. The cost of emergency contraception: results from a nationwide study. July 2013. Available at: http://americansocietyforec.org/uploads/3/2/7/0/3270267/asecpricingreport.pdf. Accessed June 22, 2019.
- Mishell DR, Jr. Intrauterine devices: mechanisms of action, safety, and efficacy. 1998;58:45S-53S; quiz 70S.
- Milsom I, Andersson K, Jonasson K, Lindstedt G, Rybo G. The influence of the Gyne-T 380S IUD on menstrual blood loss and iron status. 1995;52:175-179.
- Creinin MD, Schlaff W, Archer DF, et al. Progesterone receptor modulator for emergency contraception: a randomized controlled trial. Obstet Gynecol.2006;108:1089-1097.
- Glasier AF, Cameron ST, Fine PM, et al. Ulipristal acetate versus levonorgestrel for emergency contraception: a randomised non-inferiority trial and meta-analysis. 2010;375:555-562.
- Piaggio G, Kapp N, von Hertzen H. Effect on pregnancy rates of the delay in the administration of levonorgestrel for emergency contraception: a combined analysis of four WHO trials. 2011;84:35-39.
- Webb AM, Russell J, Elstein M. Comparison of Yuzpe regimen, danazol, and mifepristone (RU486) in oral postcoital contraception. 1992;305:927-931.
- Wanggren K, Stavreus-Evers A, Olsson C, Andersson E, Gemzell-Danielsson K. Regulation of muscular contractions in the human fallopian tube through prostaglandins and progestagens.Hum Reprod.2008;23:2359-2368.
- Wagner BL, Pollio G, Giangrande P, et al. The novel progesterone receptor antagonists RTI 3021-012 and RTI 3021-022 exhibit complex glucocorticoid receptor antagonist activities: implications for the development of dissociated antiprogestins. 1999;140:1449-1458.
- Blithe DL, Nieman LK, Blye RP, Stratton P, Passaro M. Development of the selective progesterone receptor modulator CDB-2914 for clinical indications. 2003;68:1013-1017.
- Mozzanega B, Gizzo S, Di Gangi S, Cosmi E, Nardelli GB. Ulipristal acetate: critical review about endometrial and ovulatory effects in emergency contraception. Reprod Sci.2014;21:678-685.
- Rosato E, Farris M, Bastianelli C. Mechanism of action of uliprstal acetate for emergency contraception: a systematic review. Front Pharmacol. 2015;6:315.
- Brache V, Alvarez F, Faundes A, Cochon L, Thevenin F. Effect of preovulatory insertion of Norplant implants over luteinizing hormone secretion and follicular development. Fertil Steril.1996;65:1110-1114.
- Brache V, Cochon L, Deniaud M, Croxatto HB. Ulipristal acetate prevents ovulation more effectively than levonorgestrel: analysis of pooled data from three randomized trials of emergency contraception regimens. 2013;88:611-618.
- Ella® [package insert]. Paris, France: Afaxys Pharma LLC; 2018. Available at: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=052bfe45-c485-49e5-8fc4-51990b2efba4. Accessed June 22, 2019.
- Salcedo J, Rodriguez MI, Curtis KM, Kapp N. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. 2013;87:602-604.
- Pohl O, Osterloh I, Gotteland JP. Effects of erythromycin at steady-state concentrations on the pharmacokinetics of ulipristal acetate. J Clin Pharm Ther.2013;38:512-517.
- Levy DP, Jager M, Kapp N, Abitbol JL. Ulipristal acetate for emergency contraception: postmarketing experience after use by more than 1 million women. Contraception. 2014;89:431-433.
- Kapp N, Abitbol JL, Mathe H, et al. Effect of body weight and BMI on the efficacy of levonorgestrel emergency contraception. 2015;91:97-104.
- Office of Population Research. Effectiveness: are emergency contraceptive pills effective for overweight or obese women? February 2019. Available at: http://ec.princeton.edu/questions/ecobesity.html. Accessed June 22, 2019.
- Moreau C, Trussell J. Results from pooled Phase III studies of ulipristal acetate for emergency contraception. 2012;86:673-680.
- Glasier A, Cameron ST, Blithe D, et al. Can we identify women at risk of pregnancy despite using emergency contraception? Data from randomized trials of ulipristal acetate and levonorgestrel. 2011;84:363-367.
- European Medicines Agency. Levonorgestrel and ulipristal remain suitable emergency contraceptives for all women, regardless of bodyweight. September 2014. Available at: http://www.ema.europa.eu/docs/en_GB/document_library/Referrals_document/Emergency_contraceptives_31/WC500176381.pdf. Accessed June 22, 2019.
- Merki-Feld GS, Skouby S, Serfaty D, et al. European society of contraception statement on contraception in obese women. Eur J Contracept Reprod Health Care.2015;20:19-28.
- Kohn JE, Lopez PM, Simons HR. Weight and body mass index among female contraceptive clients. 2015;91:470-473.
- von Hertzen H, Piaggio G, Ding J, et al. Low dose mifepristone and two regimens of levonorgestrel for emergency contraception: a WHO multicentre randomised trial. 2002;360:1803-1810.
- Task Force on Postovulatory Methods of Fertility R. Randomised controlled trial of levonorgestrel versus the Yuzpe regimen of combined oral contraceptives for emergency contraception. Task Force on Postovulatory Methods of Fertility Regulation. 1998;352:428-433.
- Li HW, Lo SS, Ho PC. Emergency contraception. Best Pract Res Clin Obstet Gynaecol.2014;28:835-844.
- Noe G, Croxatto HB, Salvatierra AM, et al. Contraceptive efficacy of emergency contraception with levonorgestrel given before or after ovulation. 2011;84:486-492.
- Noe G, Croxatto HB, Salvatierra AM, et al. Contraceptive efficacy of emergency contraception with levonorgestrel given before or after ovulation. Contraception. 2010;81:414-420.
- Piaggio G, von Hertzen H, Grimes DA, Van Look PF. Timing of emergency contraception with levonorgestrel or the Yuzpe regimen. Task Force on Postovulatory Methods of Fertility Regulation. 1999;353:721.
- Sanchez-Borrego R, Balasch J. Ethinyl oestradiol plus dl-norgestrel or levonorgestrel in the Yuzpe method for post-coital contraception: results of an observational study. Hum Reprod. 1996;11:2449-2453.
- De Santis M, Cavaliere AF, Straface G, Carducci B, Caruso A. Failure of the emergency contraceptive levonorgestrel and the risk of adverse effects in pregnancy and on fetal development: an observational cohort study. Fertil Steril.2005;84:296-299.
- Zhang L, Chen J, Wang Y, Ren F, Yu W, Cheng L. Pregnancy outcome after levonorgestrel-only emergency contraception failure: a prospective cohort study. Hum Reprod. 2009;24:1605-1611.
- Zhang L, Ye W, Yu W, Cheng L, Shen L, Yang Z. Physical and mental development of children after levonorgestrel emergency contraception exposure: a follow-up prospective cohort study. Biol Reprod.2014;91:27.
- Cleland K, Raymond E, Trussell J, Cheng L, Zhu H. Ectopic pregnancy and emergency contraceptive pills: a systematic review. Obstet Gynecol.2010;115:1263-1266.
- Ho PC, Kwan MS. A prospective randomized comparison of levonorgestrel with the Yuzpe regimen in post-coital contraception. Hum Reprod.1993;8:389-392.
- Raymond E, Taylor D, Trussell J, Steiner MJ. Minimum effectiveness of the levonorgestrel regimen of emergency contraception. 2004;69:79-81.
- Office of Population Research & Association of Reproductive Health Professionals. Combined emergency contraceptive pills (“morning after pills”). Available at: http://ec.princeton.edu/info/combecp.html. Accessed September 4, 2015.
- Trussell J, Jordan B. Mechanism of action of emergency contraceptive pills. 2006;74:87-89.
- Van Santen MR, Haspels AA. A comparison of high-dose estrogens versus low-dose ethinylestradiol and norgestrel combination in postcoital interception: a study in 493 women. Fertil Steril. 1985;43:206-213.
- Raymond EG, Creinin MD, Barnhart KT, Lovvorn AE, Rountree RW, Trussell J. Meclizine for prevention of nausea associated with use of emergency contraceptive pills: a randomized trial. Obstet Gynecol.2000;95:271-277.
- Rodriguez MI, Godfrey EM, Warden M, Curtis KM. Prevention and management of nausea and vomiting with emergency contraception: a systematic review. Contraception. 2013;87:583-589.
- Office of Population Research & Association of Reproductive Health Professionals. Side effects: what are the side effects of emergency contraceptive pills? Available at: http://ec.princeton.edu/questions/ecsideeffects.html. Accessed September 4, 2015.
- Group ECW. Venous thromboembolism in women: a specific reproductive health risk. Hum Reprod Update.2013;19:471-482.
- Benagiano G, Bastianelli C, Farris M, Brosens I. Selective progesterone receptor modulators: an update. Expert Opin Pharmacother. 2014;15:1403-1415.
- Carbonell JL, Garcia R, Gonzalez A, Breto A, Sanchez C. Mifepristone 5 mg versus 10 mg for emergency contraception: double-blind randomized clinical trial. Int J Womens Health.2015;7:95-102.
- Guttmacher Institute. State laws and policies: emergency contraception. June 2019. Available at: https://www.guttmacher.org/state-policy/explore/emergency-contraception. Accessed June 22, 2019.
- Polis CB, Schaffer K, Blanchard K, Glasier A, Harper CC, Grimes DA. Advance provision of emergency contraception for pregnancy prevention (full review). Cochrane Database Syst Rev. 2007:CD005497.
- Raymond EG, Trussell J, Polis CB. Population effect of increased access to emergency contraceptive pills: a systematic review. Obstet Gynecol. 2007;109:181-188.
- Rodriguez MI, Curtis KM, Gaffield ML, Jackson E, Kapp N. Advance supply of emergency contraception: a systematic review. Contraception. 2013;87:590-601.
- Raymond EG, Weaver MA. Effect of an emergency contraceptive pill intervention on pregnancy risk behavior. 2008;77:333-336.
- Gold MA, Wolford JE, Smith KA, Parker AM. The effects of advance provision of emergency contraception on adolescent women’s sexual and contraceptive behaviors. J Pediatr Adolesc Gynecol. 2004;17:87-96.
- Raine TR, Harper CC, Rocca CH, et al. Direct access to emergency contraception through pharmacies and effect on unintended pregnancy and STIs: a randomized controlled trial. JAMA. 2005;293:54-62.
- Raymond EG, Stewart F, Weaver M, Monteith C, Van Der Pol B. Impact of increased access to emergency contraceptive pills: a randomized controlled trial. Obstet Gynecol.2006;108:1098-1106.
- Ekstrand M, Larsson M, Darj E, Tyden T. Advance provision of emergency contraceptive pills reduces treatment delay: a randomised controlled trial among Swedish teenage girls. Acta Obstet Gynecol Scand.2008;87:354-359.
- Mulligan K. Access to emergency contraception and its impact on fertility and sexual behavior. Health Econ. 2015;25:455-469.
- Atkins DN, Bradford WD. Association between increased emergency contraception availability and risky sexual practices. Health Serv Res.2015;50:809-829.
- Yen S, Parmar DD, Lin EL, Ammerman S. Emergency contraception pill awareness and knowledge in uninsured adolescents: High rates of misconceptions concerning indications for use, side effects, and access. J Pediatr Adolesc Gynecol.2015;28:337-342.
- Abbott J, Feldhaus KM, Houry D, Lowenstein SR. Emergency contraception: what do our patients know? Ann Emerg Med. 2004;43:376-381.
- Ahern R, Frattarelli LA, Delto J, Kaneshiro B. Knowledge and awareness of emergency contraception in adolescents. J Pediatr Adolesc Gynecol. 2010;23:273-278.
- Ragland D, Battle M, Kueter TJ, Payakachat N. Consumer attitudes towards and satisfaction with emergency contraception counselling: experience from clinic and retail pharmacy settings. Int J Pharm Pract.2015;23:349-352.
- Lopez LM, Steiner M, Grimes DA, Hilgenberg D, Schulz KF. Strategies for communicating contraceptive effectiveness. Cochrane Database Syst Rev.2013;4:CD006964.
- Thompson K, Belden P. Counseling for emergency contraception: time for a tiered approach. 2014;90:559-561.
- Raymond EG, Halpern V, Lopez LM. Pericoital oral contraception with levonorgestrel: a systematic review. Obstet Gynecol.2011;117:673-681.
- Donnez J, Tatarchuk TF, Bouchard P, et al. Ulipristal acetate versus placebo for fibroid treatment before surgery. N Engl J Med.2012;366:409-420.
- Michie L, Cameron ST, Glasier A, Larke N, Muir A, Lorimer A. Pharmacy-based interventions for initiating effective contraception following the use of emergency contraception: a pilot study. Contraception. 2014;90:447-453.
- Schwarz EB, Papic M, Parisi SM, Baldauf E, Rapkin R, Updike G. Routine counseling about intrauterine contraception for women seeking emergency contraception. Contraception. 2014;90:66-71.
- Office of Population Research. Safety: do emergency contraceptive pills increase my risk of having an ectopic pregnancy (that’s when the pregnancy is outside the uterus)? Available at: http://ec.princeton.edu/questions/ecectopic.html. Accessed June 22, 2019.
Back to Top